 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,1′-(Pyridine-2,6-diyl)di(ethan-1-one) | |
| Other names
1,1′-(Pyridine-2,6-diyl)diethanone 1-(6-Acetylpyridin-2-yl)ethanone DAP 2,6-Bisacetylpyridine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.013.130 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
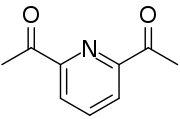
| C9H9NO2 | |
| Molar mass | 163.176 g·mol−1 |
| Appearance | White crystals |
| Density | 1.119 g/cm3 |
| Melting point | 81 °C (178 °F; 354 K) Sublimes at 110 to 130 °C (230 to 266 °F; 383 to 403 K) |
| Boiling point | 126 °C (259 °F; 399 K) |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| Safety data sheet (SDS) | MSDS sheet |
| Related compounds | |
Related pyridines |
2-acetylpyridine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
2,6-Diacetylpyridine is an organic compound with the formula C5H3N(C(O)CH3)2. It is a white solid that is soluble in organic solvents. It is a disubstituted pyridine. It is a precursor to ligands in coordination chemistry.[1][2]
Synthesis
The synthesis of 2,6-diacetylpyridine begins with oxidation of the methyl groups in 2,6-lutidine to form dipicolinic acid. This process has been well established with potassium permanganate and selenium dioxide.[3] The diketone can be formed from the diester of picolinic acid groups through a Claisen condensation.[4] The resulting adduct can be decarboxylated to give diacetylpyridine.[5]
 DAP synthesis Claisen
DAP synthesis Claisen
Treating 2,6-pyridinedicarbonitrile with methylmagnesium bromide provides an alternative synthesis for the diketone.[2]
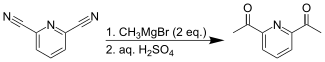 DAP synthesis Grignard
DAP synthesis Grignard
Precursor to Schiff base ligands
Diacetylpyridine is a popular starting material for ligands in coordination chemistry, often via template reactions. The diiminopyridine (DIP) class of ligands can be formed from diacetylpyridine through Schiff base condensation with substituted anilines. Diiminopyridine ligands have been the focus of much interest due to their ability to traverse a wide range of oxidation states.[2]
 diiminopyridine_synthesis
diiminopyridine_synthesis
In azamacrocycle chemistry, diacetylpyridines can undergo the same Schiff base condensation with N1-(3-aminopropyl)propane-1,3-diamines. The product of the condensation can be hydrogenated to yield macrocyclic tetradentate ligands. Similar penta- and hexadentate ligands have been synthesized by varying the polyamine chain.[1]
See also
References
- 1 2 Curtis, N. F. (2012). "The Advent of Macrocyclic Chemistry". Supramolecular Chemistry. 24 (7): 439–447. doi:10.1080/10610278.2012.688123. S2CID 96660708.
- 1 2 3 Schmidt, R.; Welch, M.B.; Palackal, S.J.; Alt, H.G. (2001). "Hydrogenized iron(II) complexes as highly active ethene polymerization catalysts". Journal of Molecular Catalysis A: Chemical. 179 (1–2): 155–173. doi:10.1016/S1381-1169(01)00333-8.
- ↑ CA patent 1108617, Agnese, G. & Burshchi, E., "Two Stage Process for Preparing 2,6-pyridinedicarboxylic acid"
- ↑ Darmon, Jonathan M.; Turner, Zoë R.; Lobkovsky, Emil; Chirik, Paul J.; Finkelstein, K. D.; Wieghardt, K.; Debeer, S.; Chirik, P. J. (2012). "Electronic Effects in 4-Substituted Bis(imino)pyridines and the Corresponding Reduced Iron Compounds". Organometallics. 31 (6): 2275–2285. doi:10.1021/om201212m. PMC 3366276. PMID 22675236.
- ↑ Yoshiro Ogata; Masaru Tsuchida; Akihiko Muramoto (2006). "Controlled Synthesis of 2-Acetyl-6-carbethioxypyridine and 2-6-Diacetylpyridine from 2,6-Dimethylpyridine". Synth. Commun. 35 (17): 2317–2324. doi:10.1080/00397910500186995. S2CID 93168188.