 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
[1,1′-Biphenyl]-3,3′,4,4′-tetramine | |
| Other names
[1,1′-Biphenyl]-3,3′,4,4′-tetraamine (not recommended) 3,3′,4,4′-Biphenyltetramine 3,3′,4,4′-Tetraamino-diphenyl | |
| Identifiers | |
3D model (JSmol) |
|
| 1212988 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.919 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII |
|
| UN number | 2811 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H14N4 C12H18Cl4N4 (4HCl) | |
| Molar mass | 214.27 g/mol 360.11 g/mol (4HCl) |
| Melting point | 175 to 177 °C (347 to 351 °F; 448 to 450 K) (280 °C for 4HCl.2H2O) |
| Hazards | |
| GHS labelling: | |
 | |
| Danger | |
| H341, H350 | |
| P201, P202, P281, P308+P313, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
mouse, oral Acute: 1834 mg/kg. |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
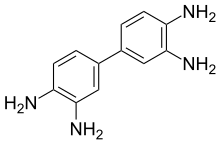
3,3′-Diaminobenzidine (DAB) is an organic compound with the formula (C6H3(NH2)2)2. This derivative of benzidine is a precursor to polybenzimidazole, which forms fibers that are renowned for their chemical and thermal stability.[1] As its water-soluble tetrahydrochloride, DAB has been used in immunohistochemical staining of nucleic acids and proteins.[2]
Structure
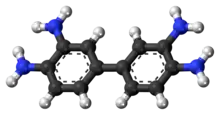
DAB is symmetric about the central carbon bond between both ring structures. In the crystal, the rings of each molecule are co-planar and the amine units connect molecules to form an intermolecular 3-dimensional hydrogen bond network.[3]
Preparation
Diaminobenzidine, which is commercially available, is prepared by treating 3,3′-dichlorobenzidine with ammonia with a copper catalyst at high temperature and pressure, followed by acidic workup.[4]
An alternate synthesis route involves the diacylation of benzidine with acetic anhydride under basic conditions:[1]
- (NH2)C6H4C6H4(NH2) + 2 (CH3CO)2O ⟶ (NHCOCH3)C6H4C6H4(NHCOCH3) + 2 CH3CO2H
The diacetylated compound then undergoes nitration with nitric acid to produce an ortho-dinitro compound due to the ortho-directing acetyl substituents:[1]
- (NHCOCH3)C6H4C6H4(NHCOCH3) + 2HNO3 ⟶ (O2N)(NHCOCH3)C6H3C6H3(NHCOCH3)(NO2) + 2H2O
The acetyl groups are then removed through saponification:[1]
- (O2N)(NHCOCH3)C6H3C6H3(NHCOCH3)(NO2) + 2NaOH ⟶ (O2N)(NH2)C6H3C6H3(NH2)(NO2) + 2(NaOCOCH3)
The dinitrobenzidine compound is then reduced with hydrochloric acid and iron to produce 3,3′-diaminobenzidine:[1]
- 3(O2N)(NH2)C6H3C6H3(NH2)(NO2) + 12HCl + 10Fe0 ⟶ 3(NH2)2C6H3C6H3(NH2)2 + 4Fe2O3 + 6FeCl2
The reduction of the dinitrobenzidine compound can also proceed with tin(II) chloride instead of iron powder or with sodium dithionite in methanol.[1]
Applications
In its main application, DAB is the precursor to polybenzimidazole.
Diaminobenzidine is oxidized by hydrogen peroxide in the presence of hemoglobin to give a dark-brown color. This color change is used to detect fingerprints in blood.[5] The solubility of DAB in water allows for adaptability compared to other detection solutions which use toxic solvents.[6] Improperly prepared tissue samples may give false positives.[7] In research, this reaction is used to stain cells that were prepared with hydrogen peroxidase enzyme, following common immunocytochemistry protocols. Relevant to Alzheimer's disease, Aβ protein amyloid plaques are targeted by a primary antibody, and subsequently by a secondary antibody, which is conjugated with a peroxidase enzyme. This will bind DAB as a substrate and oxidize it, producing an easily observable brown color. Plaques can then be quantified for further evaluation.[8] One other method uses complexes of injected biocytin with avidin or streptavidin, biotin, and then peroxidase.
References
- 1 2 3 4 5 6 Hans Schwenecke, Dieter Mayer "Benzidine and Benzidine Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim.
- ↑ Roschzttardtz, H.; Grillet, L.; Isaure, M.-P.; Conejero, G.; Ortega, R.; Curie, C.; Mari, S. (2011), "The Plant Cell Nucleolus as a Hot Spot for Iron", The Journal of Biological Chemistry, 286 (32): 27863–27866, doi:10.1074/jbc.C111.269720, PMC 3151030, PMID 21719700
- ↑ Hui-Fen Qian & Wei Huang (2010). "Biphenyl-3,3′,4,4′-tetraamine". Acta Crystallographica. E66 (5): o1060. doi:10.1107/S1600536810012511. PMC 2979072. PMID 21579117.
- ↑ US 3943175, Druin, Melvin L. & Oringer, Kenneth, "Synthesis of pure 3,3′-diaminobenzidine", issued 1976-03-09, assigned to Celanese Corporation
- ↑ "D.A.B. (Diaminobenzidine)". Chesapeake Bay Division, International Association for Identification. Archived from the original on 23 December 2007. Retrieved 2007-11-09.
- ↑ Sahs P (1992), "DAB: An Advancement in Blood Print Detection", J. Forensic Ident., 42: 412–420
- ↑ "Liquid DAB Substrate" (pdf). Dako. Retrieved 2008-02-02.
- ↑ Falangola, M. F.; Lee, S. P.; Nixon, R. A.; Duff, K. & Helpern, J.K. (2005). "Histological co-localization of iron in Abeta plaques of PS/APP transgenic mice". Neurochemical Research. 30 (2): 201–205. doi:10.1007/s11064-004-2442-x. PMC 3959869. PMID 15895823.
