 | |
 | |
| Names | |
|---|---|
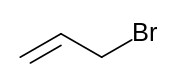
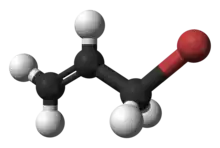
| Preferred IUPAC name
3-Bromoprop-1-ene | |
| Other names
Allyl bromide 3-Bromopropene 3-Bromopropylene 3-Bromo-1-propene Bromoallylene 2-Propenyl bromide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.003.134 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1099 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H5Br | |
| Molar mass | 120.977 g·mol−1 |
| Appearance | Clear to light yellow liquid |
| Odor | Unpleasant, irritating, pungent |
| Density | 1.398 g/cm3 |
| Melting point | −119 °C (−182 °F; 154 K) |
| Boiling point | 71 °C (160 °F; 344 K) |
| 0.38 g/100 g H2O [1] | |
| log P | 1.79[1] |
| Vapor pressure | 18.6 kPa |
| −58.6·10−6 cm3·mol−1[1] | |
Refractive index (nD) |
1.4697 (20 °C, 589.2 nm) |
| Viscosity | 0.471 cP[1] |
| ≈1.9 D[1] | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
12.2 kJ·mol−1 (liquid) 45.2 kJ·mol−1 (gas)[1] |
Enthalpy of vaporization (ΔfHvap) |
32.73 kJ·mol−1[1] |
| Hazards | |
| GHS labelling: | |
     | |
| Danger | |
| H225, H301, H314, H330, H331, H340, H350, H400 | |
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P273, P280, P281, P284, P301+P310, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P311, P320, P321, P330, P363, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | −2 to −1 °C |
| 280 °C (536 °F; 553 K) | |
| Explosive limits | 4.3–7.3 % |
Threshold limit value (TLV) |
0.1 ppm[1] (TWA), 0.2 ppm[1] (STEL) |
| Safety data sheet (SDS) | MSDS at Oxford University |
| Related compounds | |
Related compounds |
Allyl chloride Allyl iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Allyl bromide (3-bromopropene) is an organic halide. It is an alkylating agent used in synthesis of polymers, pharmaceuticals,[2] synthetic perfumes[3] and other organic compounds. Physically, allyl bromide is a colorless liquid with an irritating and persistent smell, however, commercial samples are yellow or brown.[4] Allyl bromide is more reactive but more expensive than allyl chloride, and these considerations guide its use.[5]
Preparation
Allyl bromide is produced commercially from allyl alcohol and hydrobromic acid:[5]
- CH2=CHCH2OH + HBr → CH2=CHCH2Br + H2O
It can also be prepared by the halogen-exchange reaction between allyl chloride and hydrobromic acid or by the allylic bromination of propene.[5]
Reactions and uses
Electrophilic properties
Allyl bromide is an electrophilic alkylating agent.[4] It reacts with nucleophiles, such as amines, carbanions, alkoxides, etc., to introduce the allyl group:
- CH2=CHCH2Br + Nu− → CH2=CHCH2Nu + Br− (Nu− is a nucleophile)
It is used in the synthesis of compounds containing the allyl functionality, such as the pharmaceuticals methohexital, secobarbital and thiamylal.[2]
Preparation of Grignard reagent
Allyl bromide reacts with magnesium metal in dry ether to form allylmagnesium bromide, a Grignard reagent:[6]
- CH2=CHCH2Br + Mg → CH2=CHCH2MgBr
References
- 1 2 3 4 5 6 7 8 9 CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data. William M. Haynes, David R. Lide, Thomas J. Bruno (2016-2017, 97th ed.). Boca Raton, Florida. 2016. ISBN 978-1-4987-5428-6. OCLC 930681942.
{{cite book}}: CS1 maint: location missing publisher (link) CS1 maint: others (link) - 1 2 Yoffe, David; Frim, Ron; Ukeles, Shmuel D.; Dagani, Michael J.; Barda, Henry J.; Benya, Theodore J.; Sanders, David C. (2013-10-09), "Bromine Compounds", in Wiley-VCH Verlag GmbH & Co. KGaA (ed.), Ullmann's Encyclopedia of Industrial Chemistry, Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA, pp. 1–31, doi:10.1002/14356007.a04_405.pub2, ISBN 978-3-527-30673-2, retrieved 2022-03-04
- ↑ PubChem. "Hazardous Substances Data Bank (HSDB) : 622". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-03-04.
- 1 2 Bolton, Roger (2001-04-15), "Allyl Bromide", in John Wiley & Sons, Ltd (ed.), Encyclopedia of Reagents for Organic Synthesis, Chichester, UK: John Wiley & Sons, Ltd, pp. ra045, doi:10.1002/047084289x.ra045, ISBN 978-0-471-93623-7, retrieved 2022-03-04
- 1 2 3 Dagani, M. J.; Barda, H. J.; Benya, T. J.; Sanders, D. C. "Bromine Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a04_405. ISBN 978-3527306732.
- ↑ "6-Chloro-1-Hexene and 8-Chloro-1-Octene". Organic Syntheses. 76: 221. 1999. doi:10.15227/orgsyn.076.0221.
