 | |
| Names | |
|---|---|
| Preferred IUPAC name
([1,1′-Binaphthalene]-2,2′-diyl)bis(diphenylphosphane) | |
| Other names
BINAP | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.114.880 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C44H32P2 | |
| Molar mass | 622.688 g·mol−1 |
| Appearance | Colorless solid |
| Melting point | 239 to 241 °C (462 to 466 °F; 512 to 514 K) (R) 238–240 °C (S) |
| organic solvents | |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335, H413 | |
| P261, P264, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
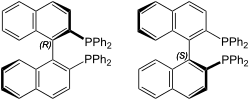
BINAP (2,2′-bis(diphenylphosphino)-1,1′-binaphthyl) is an organophosphorus compound. This chiral diphosphine ligand is widely used in asymmetric synthesis. It consists of a pair of 2-diphenylphosphinonaphthyl groups linked at the 1 and 1′ positions. This C2-symmetric framework lacks a stereogenic atom, but has axial chirality due to restricted rotation (atropisomerism). The barrier to racemization is high due to steric hindrance, which limits rotation about the bond linking the naphthyl rings. The dihedral angle between the naphthyl groups is approximately 90°. The natural bite angle is 93°.[1]
Use as ligand in asymmetric catalysis
BINAP is used in organic synthesis for enantioselective transformations catalyzed by its complexes of ruthenium, rhodium, and palladium.[2] As pioneered by Ryōji Noyori and his co-workers, rhodium complexes of BINAP are useful for the synthesis of (–)-menthol.[3][4]
Silver complexes are also important; BINAP-AgF can be used to enantioselectively protonate silyl enol ethers.[5]
Subsequent studies revealed that related diphosphines with a narrower dihedral angle between the aromatic faces give catalysts that are more enantioselective. One such ligand is SEGPHOS.[6]
Preparation
BINAP is prepared from BINOL[7][8] via its bistriflate derivatives. Both the (R)- and (S)-enantiomers, as well as the racemate, are commercially available. One of the wide applications include chemoselective hydrogenation, where BINAP is conjugated to rhodium.
Further reading
- Berthod, Mikaël; Mignani, Gérard; Woodward, Gary; Lemaire, Marc (2005). "Modified BINAP: The How and the Why". Chemical Reviews. 105 (5): 1801–1836. doi:10.1021/cr040652w. PMID 15884790.
- Genet, Jean-Pierre; Ayad, Tahar; Ratovelomanana-Vidal, Virginie (2014). "Electron-Deficient Diphosphines: The Impact of DIFLUORPHOS in Asymmetric Catalysis". Chemical Reviews. 114 (5): 2824–2880. doi:10.1021/cr4003243. PMID 24517862.
- Kočovský, Pavel; Vyskočil, Štěpán; Smrčina, Martin (2003). "Non-Symmetrically Substituted 1,1'-Binaphthyls in Enantioselective Catalysis". Chemical Reviews. 103 (8): 3213–3246. doi:10.1021/cr9900230. PMID 12914496.
- Zhang, Zhenfeng; Butt, Nicholas A.; Zhang, Wanbin (2016). "Asymmetric Hydrogenation of Nonaromatic Cyclic Substrates". Chemical Reviews. 116 (23): 14769–14827. doi:10.1021/acs.chemrev.6b00564. PMID 27960269.
- Clevenger, Andrew L.; Stolley, Ryan M.; Aderibigbe, Justis; Louie, Janis (2020). "Trends in the Usage of Bidentate Phosphines as Ligands in Nickel Catalysis". Chemical Reviews. 120 (13): 6124–6196. doi:10.1021/acs.chemrev.9b00682. PMID 32491839. S2CID 219311508.
References
- ↑ Birkholz (née Gensow), Mandy-Nicole; Freixa, Zoraida; van Leeuwen, Piet W. N. M. (2009). "Bite angle effects of diphosphines in C–C and C–X bond forming cross coupling reactions". Chemical Society Reviews. 38 (4): 1099–1118. doi:10.1039/B806211K. PMID 19421583.
- ↑ Kitamura, Masato; M. Tokunaga; T. Ohkuma; R. Noyori (1998). "Asymmetric hydrogenation of 3-oxo carboxylates using BINAP-ruthenium complexes". Org. Synth. 9: 589.
- ↑ Akutagawa, S (1992). "A practical synthesis of (−)-menthol with the Rh-BINAP catalyst". Chirality Ind.: 313–323.
- ↑ Kumobayashi, Hidenori; Sayo, Noboru; Akutagawa, Susumu; Sakaguchi, Toshiaki; Tsuruta, Haruki (1997). "Industrial asymmetric synthesis by use of metal-BINAP catalysts". Nippon Kagaku Kaishi. 12 (12): 835–846. doi:10.1246/nikkashi.1997.835.
- ↑ Yanagisawa, Akira; Touge, Taichiro; Takayoshi, Arai (2005). "Enantioselective Protonation of Silyl Enolates Catalyzed by a Binap⋅AgF Complex". Angewandte Chemie International Edition. 44 (10): 1546–8. doi:10.1002/anie.200462325. PMID 15645475.
- ↑ Shimizu H, Nagasaki I, Matsumura K, Sayo N, Saito T (2007). "Developments in Asymmetric Hydrogenation from an Industrial Perspective". Acc. Chem. Res. 40 (12): 1385–1393. doi:10.1021/ar700101x. PMID 17685581.
- ↑ "BINAP: An industrial approach to manufacture" (PDF). Rhodia. Retrieved 2008-10-20.
- ↑ Cai, Dongwei; J. F. Payack; D. R. Bender; D. L. Hughes; T. R. Verhoeven; P. J. Reider (1999). "(R)-(+)- and (S)-(−)-2,2′-bis(diphenylphosphino)-1,1′-binapthyl (BINAP)". Organic Syntheses. 76: 6.