| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Oxepan-2-one | |||
| Other names
Caprolactone ε-Caprolactone Hexano-6-lactone 6-Hexanolactone Hexan-6-olide 1-Oxa-2-oxocycloheptane | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.007.217 | ||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C6H10O2 | |||
| Molar mass | 114.14 g/mol | ||
| Density | 1.030 g/cm3 | ||
| Melting point | −1 °C (30 °F; 272 K) | ||
| Boiling point | 241 °C (466 °F; 514 K)[1] | ||
| Miscible [2] | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
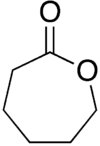
ε-Caprolactone or simply caprolactone is a lactone (a cyclic ester) possessing a seven-membered ring. Its name is derived from caproic acid. This colorless liquid is miscible with most organic solvents and water. It was once produced on a large scale as a precursor to caprolactam.[3]
Production and uses
Caprolactone is prepared industrially by Baeyer-Villiger oxidation of cyclohexanone with peracetic acid.
Caprolactone is a monomer used in the production of highly specialised polymers. Ring-opening polymerization, for example, gives polycaprolactone.[3] Another polymer is polyglecaprone, used as suture material in surgery.[4]
Reactions
Although no longer economical, caprolactone was once produced as a precursor to caprolactam. Caprolactone is treated with ammonia at elevated temperatures to give the lactam:
- (CH2)5CO2 + NH3 → (CH2)5C(O)NH + H2O
Carbonylation of caprolactone gives, after hydrolysis, pimelic acid. The lactone ring is easily opened with nucleophiles including alcohols and water to give polylactones and eventually the 6-hydroxyadipic acid.
Related compounds
Several other caprolactones are known, including α-, β-, γ-, and δ-caprolactones. All are chiral. (R)-γ-caprolactone is a component of floral scents and of the aromas of some fruits and vegetables,[5] and is also produced by the Khapra beetle as a pheromone.[6] δ-caprolactone is found in heated milk fat.[7]
An ether of caprolactone is used as a binder for AP/AN/Al rocket propellant HTCE: Hydroxy-Terminated Caprolactone Ether [8]
Safety
Caprolactone hydrolyses rapidly and the resulting hydroxycarboxylic acid displays unexceptional toxicity, as is common for the other hydroxycarboxylic acids.[9] It is known to cause severe eye irritation. Exposure may result in corneal injury.[2]
References
- ↑ "Capa Monomer product data sheet" (PDF). Perstorp. 2015-02-27. Archived from the original (PDF) on 2017-02-02. Retrieved 2017-10-11.
- 1 2 "ε-caprolactone SIDS Initial Assessment Report" (PDF). OECD. Archived from the original (PDF) on 2011-08-15. Retrieved 2017-10-12.
- 1 2 Köpnick, Horst; Schmidt, Manfred; Brügging, Wilhelm; Rüter, Jörn; Kaminsky, Walter (2002). "Polyesters". Ullmann's Encyclopedia of Industrial Chemistry (6th ed.). Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_227.
- ↑ "glycolide E-caprolactone copolymer Summary Report". CureHunter. Retrieved 2017-10-11.
- ↑ Mosandl, A.; Günther, C. (1989). "Stereoisomeric flavor compounds: structure and properties of gamma-lactone enantiomers". Journal of Agricultural and Food Chemistry. 37: 413–418. doi:10.1021/jf00086a031.
- ↑ Nunez, M. Teresa; Martin, Victor S. (1990). "Efficient oxidation of phenyl groups to carboxylic acids with ruthenium tetraoxide. A simple synthesis of (R)-gamma-caprolactone, the pheromone of Trogoderma granarium". Journal of Organic Chemistry. 55 (6): 1928–1932. doi:10.1021/jo00293a044.
- ↑ Parliament, Thomas H.; Nawar, Wassef W.; Fagerson, Irving S. (1965). "Delta-Caprolactone in Heated Milk Fat". Journal of Dairy Science. 48 (5): 615–616. doi:10.3168/jds.S0022-0302(65)88298-4.
- ↑ HTCE
- ↑ Miltenberger, Karlheinz (2002). "Hydroxycarboxylic Acids, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry (6th ed.). Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_507.