 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Aminopropan-2-ol | |
| Other names
1-Amino-2-propanol Isopropanolamine MIPA; Threamine | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.057 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H9NO | |
| Molar mass | 75.111 g·mol−1 |
| Appearance | liquid |
| Odor | ammonia-like |
| Density | 0.973 g/mL (18 °C) [1] |
| Melting point | 1.74 °C (35.13 °F; 274.89 K) |
| Boiling point | 159.46 °C (319.03 °F; 432.61 K) |
| soluble | |
| Solubility | soluble in alcohol, ether, acetone, benzene, CCl4 |
Refractive index (nD) |
1.4479 |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 77 °C (171 °F; 350 K) |
| 374 °C (705 °F; 647 K) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
4.26 g/kg (rat, oral) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
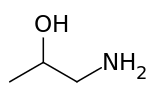
1-Aminopropan-2-ol is the organic compound with the formula CH3CH(OH)CH2NH2. It is an amino alcohol. The term isopropanolamine may also refer more generally to the additional homologs diisopropanolamine (DIPA) and triisopropanolamine (TIPA).
1-Aminopropan-2-ol is chiral. It can be prepared by the addition of aqueous ammonia to propylene oxide.[2]
Biosynthesis
(R)-1-Aminopropan-2-ol is one of the components incorporated in the biosynthesis of cobalamin. The O-phosphate ester is produced from threonine by the enzyme Threonine-phosphate decarboxylase.[3][4]
Applications
The isopropanolamines are used as buffers. They are good solubilizers of oil and fat, so they are used to neutralize fatty acids and sulfonic acid-based surfactants. Racemic 1-aminopropan-2-ol is typically used in metalworking fluid, waterborne coatings, personal care products, and in the production of titanium dioxide and polyurethanes.[5] It is an intermediate in the synthesis of a variety of pharmaceutical drugs.
(R)-1-aminopropan-2-ol is metabolised to aminoacetone by the enzyme (R)-aminopropanol dehydrogenase.[6]
Synthesis of Hexylcaine is one application.
References
- ↑ Amino-2-propanol at Sigma-Aldrich
- ↑ Smith, Michael B. (19 February 2020). March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure. John Wiley & Sons. ISBN 9781119371809.
- ↑ Cheong, Cheom-Gil; Bauer, Cary B.; Brushaber, Kevin R.; Escalante-Semerena, Jorge C.; Rayment, Ivan (2002). "Three-Dimensional Structure of the L-Threonine-O-3-phosphate Decarboxylase (CobD) Enzyme from Salmonella enterica". Biochemistry. 41 (15): 4798–4808. doi:10.1021/bi012111w. PMID 11939774.
- ↑ Warren, Martin J.; Raux, Evelyne; Schubert, Heidi L.; Escalante-Semerena, Jorge C. (2002). "The biosynthesis of adenosylcobalamin (Vitamin B12)". Natural Product Reports. 19 (4): 390–412. doi:10.1039/b108967f. PMID 12195810.
- ↑ "Monoisopropanolamine". Nanjing HBL International. Retrieved 2020-04-28.
- ↑ Turner, JM (1967). "Microbial metabolism of amino ketones. L-1-aminopropan-2-ol dehydrogenase and L-threonine dehydrogenase in Escherichia coli". Biochemical Journal. 104 (1): 112–121. doi:10.1042/bj1040112. PMC 1270551. PMID 5340733.
