Polymeric immunoglobulin receptor (pIgR) is a transmembrane protein that in humans is encoded by the PIGR gene.[5] It is an Fc receptor which facilitates the transcytosis of the soluble polymeric isoforms of immunoglobulin A and immunoglobulin M (pIg) and immune complexes. pIgRs are mainly located on the epithelial lining of mucosal surfaces of the gastrointestinal tract. The composition of the receptor is complex, including 6 immunoglobulin-like domains, a transmembrane region, and an intracellular domain.[6] pIgR expression is under the strong regulation of cytokines, hormones, and pathogenic stimuli.[6][7]
Structure
pIgR is produced among others by intestinal epithelial cells (IECs) and bronchial epithelial cells. pIgR belongs to the family of type I transmembrane proteins. The extracellular portion of the protein contains 6 domains: 5 evolutionary conserved immunoglobulin-like domains,[8] and 1 non-homologous domain, which is involved in proteolytic cleavage of pIg-pIgR complex from the apical side of the IECs. The quite long intracellular domain of the receptor, along with the transmembrane region, is responsible for the transduction of highly conserved signals.[7][9] During transcytosis, an essential part of pIgR, the secretory component, is attached to the ligand and later cleaved with the ligand to form fully functioning secreted IgA.[10]
History
Per Brandtzaeg showed that secretory component acts as a plasma membrane receptor on epithelial cells for polymeric immunoglobulin A and immunoglobulin M.[11] This was paradoxical, as secretory component is a soluble protein, whereas plasma membrane receptors are transmembrane proteins. Numerous models were proposed for how secretory component might work as a receptor, though none of these models resolved this paradox.[11] Keith Mostov and colleagues found that secretory component was a proteolytic fragment of a transmembrane precursor, the pIgR, which led them to propose the currently accepted model[12]
Function
Polymeric immunoglobulin receptor is responsible for transcytosis of soluble dimeric IgA, pentameric IgM, and immune complexes from the basolateral to the apical mucosal epithelial cell surface. pIgR has a strong specificity to polymeric immunoglobulins and is not responsive to monomeric immunoglobulin.[13] The ligand’s J-chain is responsible for the binding of pIgR to its ligand.[7][10]
Transcytosis
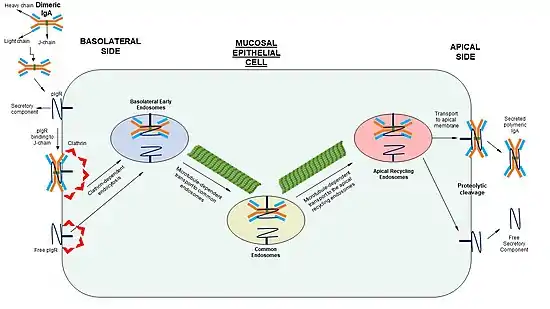
The process of transporting polymeric immunoglobulins from the basolateral to apical side, known as transcytosis, is composed of several distinct steps. Transcytosis is initiated by either the binding of dimeric IgA to the receptor or the phosphorylation of Ser-664 residue of the receptor.[9] The internalization of both free and IgA-bound pIgR is mediated by clathrin coating. The internalized receptor is transported to basolateral early endosomes. The following step of transporting the pIgR across the cell (through tubulo-vesicular compartments to apical recycling endosome) is dependent on microtubules.[9] When pIgR reaches the apical membrane, proteolytic cleavage generates either a free secretory component of SC-IgA complex , which is released to the apical lumen.[7] Cleavage occurs at the junction of the transmembrane region of the receptor and domain 5.[6]
Elimination of Immune Complexes
pIgRs are capable of capturing IgA bound to an antigen (Immune complexes (ICs)) with identical affinity as IgA and transport them to apical side. ICs result from the capture of an antigen by an antibody. IgA ICs are formed within the mucous membranes in response to foreign invasion.[14] The accumulation of ICs on the basolateral side of mucous layers can have detrimental effects. Transcytosis of IgA ICs from the formation sites represents an important mechanism of eliminating circulating antigens and minimizing their negative effects.[14][15]
Regulation
Cytokinetic Regulation
The expression of pIgR is critically regulated by the pro-inflammatory cytokines, such as IL-1, IL-4, TNF-α, and IFN-γ. The transcriptional regulation by different cytokines proceeds through similar pathways, involving the NF-kB feedback loop. Interaction of IL-1 and TNF-α with their receptors ultimately lead to transcriptional activation of PIGR gene due to nuclear translocation of NF-kB. NF-kB interacts with intron 1 of the PIGR gene to start pIgR mRNA synthesis.[6]
Besides NF-kB pathway, the transcriptional induction also proceeds in response to IFN-γ, upregulating the expression of pIgR.[6][7]
Additionally, instead of the usual antagonistic behavior, IL-4 acts synergistically with IFN-γ to induce pIgR transcription. Their combination exhibits an upregulating effect in PIGR expression because of the presence of STAT6 enhancer, the main downstream effector of IL-4, binding site in PIGR's intron 1.[16]
Hormonal Regulation
The level of pIgRs in the mucosal reproductive tract is highly dependent on the activity of sex hormones and correlates with estrous cycle phases. The peaks of pIgR expression at proestrus and estrus phases are due to the dominant activity of estrogen, which acts as a pIgR agonist. The low levels of pIgR during the diestrus are linked to the downregulating activity of progesterone, which peaks during this phase and is able to reverse the activity of estrogen.[17] Androgens are the agonists of pIgR expression in both male and female reproductive tissues.[6]
5’-flanking region of the Pigr gene contains a response element to glucocorticoids. This class of hormones increases the steady mRNA expression levels of pIgR of intestinal cells.[16][18]
Prolactin elevates the levels of IRF-1 via Jak-STAT pathway. IRF-1 is known to be a direct agonist of pIgR expression. Considering this linkage, prolactin is believed to exhibit indirect upregulation of pIgR levels during pregnancy and lactation.[6]
Pathogenic Stimulation
IECs express a variety of Toll-like receptors (TLRs), activation of which ultimately leads to the pIgR upregulation during the infection.[6][7] The most prominent modulators of pIgR regulation consist of TLR4 and TLR3, which recognize bacterial lipopolysaccharide and viral dsRNA respectively. TLR4, like the majority of TLRs, transduce the signal though MyD88 adaptor and execute the function via NF-kB, which stimulates the expression of pIgR by binding to intron 1 of the gene. TLR3, on the other hand, involves the regulation by the means of IRF-1, which is able to promote the transcription of PIGR gene by binding to exon 1.[16][18]
References
- 1 2 3 GRCh38: Ensembl release 89: ENSG00000162896 - Ensembl, May 2017
- 1 2 3 GRCm38: Ensembl release 89: ENSMUSG00000026417 - Ensembl, May 2017
- ↑ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ "Entrez Gene: PIGR polymeric immunoglobulin receptor".
- 1 2 3 4 5 6 7 8 Kaetzel CS (August 2005). "The polymeric immunoglobulin receptor: bridging innate and adaptive immune responses at mucosal surfaces". Immunological Reviews. 206: 83–99. doi:10.1111/j.0105-2896.2005.00278.x. PMID 16048543. S2CID 43588042.
- 1 2 3 4 5 6 Asano M, Komiyama K (June 2011). "Polymeric immunoglobulin receptor". Journal of Oral Science. 53 (2): 147–156. doi:10.2334/josnusd.53.147. PMID 21712618.
- ↑ Williams, Alan F. (March 1984). "Immunology: The immunoglobulin superfamily takes shape". Nature. 308 (5954): 12–13. Bibcode:1984Natur.308...12W. doi:10.1038/308012a0. ISSN 1476-4687. PMID 6700707. S2CID 4356420.
- 1 2 3 Mostov K (December 1991). "The polymeric immunoglobulin receptor". Seminars in Cell Biology. 2 (6): 411–418. PMID 1813030.
- 1 2 Owen, Judith A; Punt, Jenni; Stranford, Sharon A; Jones, Patricia P; Kuby, Janis (2013). Kuby immunology. New York: W.H. Freeman. ISBN 9781429219198. OCLC 820117219.
- 1 2 Brandtzaeg P (May 1981). "Transport models for secretory IgA and secretory IgM". Clinical and Experimental Immunology. 44 (2): 221–232. PMC 1537350. PMID 6118214.
- ↑ Mostov KE, Kraehenbuhl JP, Blobel G (December 1980). "Receptor-mediated transcellular transport of immunoglobulin: synthesis of secretory component as multiple and larger transmembrane forms". Proceedings of the National Academy of Sciences of the United States of America. 77 (12): 7257–7261. Bibcode:1980PNAS...77.7257M. doi:10.1073/pnas.77.12.7257. PMC 350481. PMID 6938972.
- ↑ Kaetzel CS, Blanch VJ, Hempen PM, Phillips KM, Piskurich JF, Youngman KR (May 1997). "The polymeric immunoglobulin receptor: structure and synthesis". Biochemical Society Transactions. 25 (2): 475–480. doi:10.1042/bst0250475. PMID 9191139.
- 1 2 Kaetzel CS, Robinson JK, Chintalacharuvu KR, Vaerman JP, Lamm ME (October 1991). "The polymeric immunoglobulin receptor (secretory component) mediates transport of immune complexes across epithelial cells: a local defense function for IgA". Proceedings of the National Academy of Sciences of the United States of America. 88 (19): 8796–8800. Bibcode:1991PNAS...88.8796K. doi:10.1073/pnas.88.19.8796. PMC 52597. PMID 1924341.
- ↑ Phalipon A, Corthésy B (February 2003). "Novel functions of the polymeric Ig receptor: well beyond transport of immunoglobulins". Trends in Immunology. 24 (2): 55–58. doi:10.1016/s1471-4906(02)00031-5. PMID 12547499.
- 1 2 3 Johansen FE, Kaetzel CS (November 2011). "Regulation of the polymeric immunoglobulin receptor and IgA transport: new advances in environmental factors that stimulate pIgR expression and its role in mucosal immunity". Mucosal Immunology. 4 (6): 598–602. doi:10.1038/mi.2011.37. PMC 3196803. PMID 21956244.
- ↑ Kaushic C, Richardson JM, Wira CR (July 1995). "Regulation of polymeric immunoglobulin A receptor messenger ribonucleic acid expression in rodent uteri: effect of sex hormones". Endocrinology. 136 (7): 2836–2844. doi:10.1210/endo.136.7.7789308. PMID 7789308.
- 1 2 Kaetzel CS (December 2014). "Cooperativity among secretory IgA, the polymeric immunoglobulin receptor, and the gut microbiota promotes host-microbial mutualism". Immunology Letters. 162 (2 Pt A): 10–21. doi:10.1016/j.imlet.2014.05.008. PMC 4246051. PMID 24877874.
Further reading
- Kaetzel CS (August 2005). "The polymeric immunoglobulin receptor: bridging innate and adaptive immune responses at mucosal surfaces". Immunological Reviews. 206: 83–99. doi:10.1111/j.0105-2896.2005.00278.x. PMID 16048543. S2CID 43588042.
- Krajci P, Kvale D, Taskén K, Brandtzaeg P (September 1992). "Molecular cloning and exon-intron mapping of the gene encoding human transmembrane secretory component (the poly-Ig receptor)". European Journal of Immunology. 22 (9): 2309–2315. doi:10.1002/eji.1830220920. PMID 1355431. S2CID 43780753.
- Krajci P, Grzeschik KH, Geurts van Kessel AH, Olaisen B, Brandtzaeg P (October 1991). "The human transmembrane secretory component (poly-Ig receptor): molecular cloning, restriction fragment length polymorphism and chromosomal sublocalization". Human Genetics. 87 (6): 642–648. doi:10.1007/BF00201717. PMID 1682231. S2CID 20403045.
- Eiffert H, Quentin E, Wiederhold M, Hillemeir S, Decker J, Weber M, Hilschmann N (February 1991). "Determination of the molecular structure of the human free secretory component". Biological Chemistry Hoppe-Seyler. 372 (2): 119–128. doi:10.1515/bchm3.1991.372.1.119. PMID 1859628.
- Bakos MA, Kurosky A, Goldblum RM (November 1991). "Characterization of a critical binding site for human polymeric Ig on secretory component". Journal of Immunology. 147 (10): 3419–3426. doi:10.4049/jimmunol.147.10.3419. PMID 1940346. S2CID 42323966.
- Krajci P, Solberg R, Sandberg M, Oyen O, Jahnsen T, Brandtzaeg P (February 1989). "Molecular cloning of the human transmembrane secretory component (poly-Ig receptor) and its mRNA expression in human tissues". Biochemical and Biophysical Research Communications. 158 (3): 783–789. doi:10.1016/0006-291X(89)92790-3. PMID 2920039.
- Davidson MK, Le Beau MM, Eddy RL, Shows TB, DiPietro LA, Kingzette M, Hanly WC (1989). "Genetic mapping of the human polymeric immunoglobulin receptor gene to chromosome region 1q31----q41". Cytogenetics and Cell Genetics. 48 (2): 107–111. doi:10.1159/000132601. PMID 3197448.
- Eiffert H, Quentin E, Decker J, Hillemeir S, Hufschmidt M, Klingmüller D, et al. (December 1984). "[The primary structure of human free secretory component and the arrangement of disulfide bonds]". Hoppe-Seyler's Zeitschrift für physiologische Chemie. 365 (12): 1489–1495. doi:10.1515/bchm2.1984.365.2.1489. PMID 6526384.
- Mizoguchi A, Mizuochi T, Kobata A (August 1982). "Structures of the carbohydrate moieties of secretory component purified from human milk". The Journal of Biological Chemistry. 257 (16): 9612–9621. doi:10.1016/S0021-9258(18)34116-4. PMID 7107583.
- Coyne RS, Siebrecht M, Peitsch MC, Casanova JE (December 1994). "Mutational analysis of polymeric immunoglobulin receptor/ligand interactions. Evidence for the involvement of multiple complementarity determining region (CDR)-like loops in receptor domain I". The Journal of Biological Chemistry. 269 (50): 31620–31625. doi:10.1016/S0021-9258(18)31739-3. PMID 7989333.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–174. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Piskurich JF, France JA, Tamer CM, Willmer CA, Kaetzel CS, Kaetzel DM (March 1993). "Interferon-gamma induces polymeric immunoglobulin receptor mRNA in human intestinal epithelial cells by a protein synthesis dependent mechanism". Molecular Immunology. 30 (4): 413–421. doi:10.1016/0161-5890(93)90071-I. PMID 8455639.
- Piskurich JF, Youngman KR, Phillips KM, Hempen PM, Blanchard MH, France JA, Kaetzel CS (January 1997). "Transcriptional regulation of the human polymeric immunoglobulin receptor gene by interferon-gamma". Molecular Immunology. 34 (1): 75–91. doi:10.1016/S0161-5890(96)00079-X. PMID 9182878.
- Hughes GJ, Frutiger S, Savoy LA, Reason AJ, Morris HR, Jaton JC (June 1997). "Human free secretory component is composed of the first 585 amino acid residues of the polymeric immunoglobulin receptor". FEBS Letters. 410 (2–3): 443–446. doi:10.1016/S0014-5793(97)00629-7. PMID 9237679.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–156. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Ackermann LW, Wollenweber LA, Denning GM (May 1999). "IL-4 and IFN-gamma increase steady state levels of polymeric Ig receptor mRNA in human airway and intestinal epithelial cells". Journal of Immunology. 162 (9): 5112–5118. doi:10.4049/jimmunol.162.9.5112. PMID 10227981. S2CID 255363351.
- Røe M, Norderhaug IN, Brandtzaeg P, Johansen FE (May 1999). "Fine specificity of ligand-binding domain 1 in the polymeric Ig receptor: importance of the CDR2-containing region for IgM interaction". Journal of Immunology. 162 (10): 6046–6052. doi:10.4049/jimmunol.162.10.6046. PMID 10229845.
- Norderhaug IN, Johansen FE, Krajci P, Brandtzaeg P (October 1999). "Domain deletions in the human polymeric Ig receptor disclose differences between its dimeric IgA and pentameric IgM interaction". European Journal of Immunology. 29 (10): 3401–3409. doi:10.1002/(SICI)1521-4141(199910)29:10<3401::AID-IMMU3401>3.0.CO;2-G. PMID 10540352.
- Mostov KE, Friedlander M, Blobel G (1984). "The receptor for transepithelial transport of IgA and IgM contains multiple immunoglobulin-like domains". Nature. 308 (5954): 37–43. Bibcode:1984Natur.308...37M. doi:10.1038/308037a0. PMID 6322002. S2CID 4340398.
- Hughes GJ, Reason AJ, Savoy L, Jaton J, Frutiger-Hughes S (September 1999). "Carbohydrate moieties in human secretory component". Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology. 1434 (1): 86–93. doi:10.1016/S0167-4838(99)00168-5. PMID 10556562.
External links
- Polymeric+Immunoglobulin+Receptor at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
This article incorporates text from the United States National Library of Medicine, which is in the public domain.