 | |||
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hexacarbonyltungsten | |||
| Other names
Tungsten carbonyl Hexacarbonylwolfram | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ECHA InfoCard | 100.034.423 | ||
| EC Number |
| ||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C6O6W | |||
| Molar mass | 351.901 g/mol | ||
| Appearance | Colorless solid | ||
| Density | 2.65 g/cm3 | ||
| Melting point | 170 °C (338 °F; 443 K) (decomposes) | ||
| insoluble | |||
| Solubility | sparingly in THF | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Flammable, CO source | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | External SDS | ||
| Related compounds | |||
Other cations |
Chromium hexacarbonyl Molybdenum hexacarbonyl | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
Tungsten hexacarbonyl (also called tungsten carbonyl) is an organometallic compound with the formula W(CO)6. This complex gave rise to the first example of a dihydrogen complex.[2]
Like its chromium and molybdenum analogs, this colorless compound is noteworthy as a volatile, air-stable derivative of tungsten in its zero oxidation state.
Preparation, properties, and structure
Like many metal carbonyls, W(CO)6 is generally prepared by "reductive carbonylation", which involves the reduction of a metal halide with under an atmosphere of carbon monoxide. As described in a 2023 survey of methods "most cost-effective routes for the synthesis of group 6 hexacarbonyls are based on the reduction of the metal chlorides (CrCl3, MoCl5 or WCl6) with magnesium, zinc or aluminium powders... under CO pressures".[3] The compound is relatively air-stable. It is sparingly soluble in nonpolar organic solvents. Tungsten carbonyl is widely used in electron beam-induced deposition technique - it is easily vaporized and decomposed by the electron beam providing a convenient source of tungsten atoms.[4]
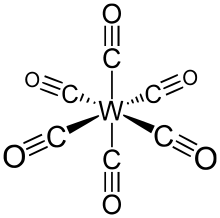
W(CO)6 adopts an octahedral geometry consisting of six rod-like CO ligands radiating from the central W atom with dipole moment 0 debye.
Reactivity
All reactions of W(CO)6 commence with displacement of some CO ligands in W(CO)6. W(CO)6 behaves similarly to the Mo(CO)6 but tends to form compounds that are kinetically more robust.
6.svg.png.webp)
Treatment of tungsten hexacarbonyl with sodium cyclopentadienide followed by oxidation of the resulting NaW(CO)3(C5H5) gives cyclopentadienyltungsten tricarbonyl dimer.[5]
One derivative is the dihydrogen complex W(CO)3[P(C6H11)3]2(H2).[2]
Three of these CO ligands can be displaced by acetonitrile.[6] W(CO)6 has been used to desulfurize organosulfur compounds and as a precursor to catalysts for alkene metathesis.
Safety and handling
Like all metal carbonyls, W(CO)6 is a dangerous source of volatile metal as well as CO.
References
- ↑ Even, J.; Yakushev, A.; Dullmann, C. E.; Haba, H.; Asai, M.; Sato, T. K.; Brand, H.; Di Nitto, A.; Eichler, R.; Fan, F. L.; Hartmann, W.; Huang, M.; Jager, E.; Kaji, D.; Kanaya, J.; Kaneya, Y.; Khuyagbaatar, J.; Kindler, B.; Kratz, J. V.; Krier, J.; Kudou, Y.; Kurz, N.; Lommel, B.; Miyashita, S.; Morimoto, K.; Morita, K.; Murakami, M.; Nagame, Y.; Nitsche, H.; et al. (2014). "Synthesis and detection of a seaborgium carbonyl complex". Science. 345 (6203): 1491–3. Bibcode:2014Sci...345.1491E. doi:10.1126/science.1255720. PMID 25237098. S2CID 206558746. (subscription required)
- 1 2 Kubas, G. J., Metal Dihydrogen and σ-Bond Complexes, Kluwer Academic/Plenum Publishers: New York, 2001
- ↑ Bruno, Sofia M.; Valente, Anabela A.; Gonçalves, Isabel S.; Pillinger, Martyn (2023). "Group 6 Carbonyl Complexes of N,O,P-Ligands as Precursors of High-Valent Metal-Oxo Catalysts for Olefin Epoxidation". Coordination Chemistry Reviews. 478: 214983. doi:10.1016/j.ccr.2022.214983. S2CID 255329673.
- ↑ Randolph, S.; Fowlkes, J.; Rack, P. (2006). "Focused, Nanoscale Electron-Beam-Induced Deposition and Etching". Critical Reviews of Solid State and Materials Sciences. 31 (3): 55. Bibcode:2006CRSSM..31...55R. doi:10.1080/10408430600930438. S2CID 93769658.
- ↑ Manning, A. R.; Hacket, Paul; Birdwhistell, Ralph (1990). "Hexacarbonylbis(η5‐Cyclopentadienyl)Dichromium, Molybdenum, and Tungsten and their Analogs, M2(η5‐C5H4R)2(CO)6 (M = Cr, Mo, and W; R = H, Me or PhCH2)". Inorganic Syntheses. 28: 148–149. doi:10.1002/9780470132593.ch39. ISBN 9780470132593.
- ↑ Kubas, G. J.; van der Sluys, L. S. (1990). "TricarbonylTris(nitrile) Complexes of Cr, Mo, and W". Inorganic Syntheses. Inorganic Syntheses. Vol. 28. pp. 29–33. doi:10.1002/9780470132593.ch6. ISBN 9780470132593.


