 | |
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Humanized |
| Target | CD19 |
| Clinical data | |
| Pronunciation | /ˌlɒnkæsˈtʌksɪmæb.ˈtɛsɪriːn/ LON-kas-TUK-si-mab TE-si-reen |
| Trade names | Zynlonta |
| Other names | ADCT-402, loncastuximab tesirine-lpyl |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a621031 |
| License data | |
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| DrugBank | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C6544H10048N1718O2064S52 |
| Molar mass | 147481.45 g·mol−1 |
Loncastuximab tesirine, sold under the brand name Zynlonta, is a monoclonal antibody conjugate medication used to treat large B-cell lymphoma and high-grade B-cell lymphoma.[1][2] It is an antibody-drug conjugate (ADC) composed of a humanized antibody targeting the protein CD19.[1]
The most common side effects include increased levels of gamma-glutamyltransferase (GGT, a liver enzyme), neutropenia (low levels of neutrophils, a type of white blood cell), tiredness, anemia (low levels of red blood cells), thrombocytopenia (low levels of blood platelets), nausea (feeling sick), peripheral edema (swelling due to fluid retention, especially of the ankles and feet) and rash.[2]
Loncastuximab tesirine was approved for medical use in the United States in April 2021,[1][3][4] and in the European Union in December 2022.[2] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication.[5]
Medical uses
Loncastuximab tesirine is indicated for the treatment of adults with relapsed or refractory large B-cell lymphoma and high-grade B-cell lymphoma.[1][2]
Technology
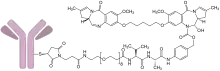
The humanized monoclonal antibody is stochastically conjugated via a valine-alanine cleavable, maleimide linker to a cytotoxic (anticancer) pyrrolobenzodiazepine (PBD) dimer. The antibody binds to CD19, a protein which is highly expressed on the surface of B-cell hematological tumors[6] including certain forms of lymphomas and leukemias. After binding to the tumor cells the antibody is internalized, the cytotoxic drug PBD is released and the cancer cells are killed. PBD dimers are generated out of PBD monomers, a class of natural products produced by various actinomycetes. PBD dimers work by crosslinking specific sites of the DNA, blocking the cancer cells’ division that cause the cells to die. As a class of DNA-crosslinking agents they are significantly more potent than systemic chemotherapeutic drugs.[7]
History
The benefit and side effects of loncastuximab tesirine were evaluated in one clinical trial, ADCT-402-201 (LOTIS-2 / NCT03589469), that included 145 participants with relapsed or refractory diffuse large B-cell lymphoma after at least two prior treatments that did not work or were no longer working.[1][4] Participants received loncastuximab tesirine 0.15 mg/kg every 3 weeks for 2 treatment cycles, then 0.075 mg/kg every 3 weeks for subsequent treatment cycles.[4] Loncastuximab tesirine treatment was continued until either disease worsened or participants experienced unacceptable side effects (toxicity).[4] The benefit of loncastuximab tesirine was evaluated by measuring how many participants had complete or partial tumor shrinkage (response) and by how long that response lasted.[4] Participants in the clinical trial were also evaluated for side effects for the purpose of this drug application.[4] Trials were conducted at 28 sites in the United States, the United Kingdom, Italy, and Switzerland.[4]
Loncastuximab tesirine was granted orphan drug designation by the FDA for the treatment of diffuse large B-cell lymphoma.[8][5] Loncastuximab tesirine was approved under FDA's accelerated approval program.[4]
Society and culture
Legal status
On 15 September 2022, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Zynlonta, intended for the treatment of adults with diffuse large B-cell lymphoma (DLBCL) and high-grade B-cell lymphoma (HGBL).[9] The applicant for this medicinal product is ADC Therapeutics (NL) B.V.[9] Loncastuximab tesirine was approved for medical use in the European Union in December 2022.[2][10]
Research
Given its mechanism of action, loncastuximab tesirine may be appealing in patients ineligible for CAR-T cell therapy.[11]
References
- 1 2 3 4 5 6 "Zynlonta- loncastuximab tesirine injection, powder, lyophilized, for solution". DailyMed. Archived from the original on 2 June 2021. Retrieved 1 June 2021.
- 1 2 3 4 5 6 "Zynlonta EPAR". European Medicines Agency (EMA). 14 September 2022. Archived from the original on 27 January 2023. Retrieved 26 January 2023. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ "Drug Approval Package: Zynlonta". U.S. Food and Drug Administration (FDA). 24 May 2021. Archived from the original on 2 June 2021. Retrieved 1 June 2021.
- 1 2 3 4 5 6 7 8 "Drug Trials Snapshots: Zynlonta". U.S. Food and Drug Administration. 23 April 2021. Archived from the original on 28 July 2023. Retrieved 9 September 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - 1 2 Advancing Health Through Innovation: New Drug Therapy Approvals 2021 (PDF). U.S. Food and Drug Administration (FDA) (Report). 13 May 2022. Archived from the original on 6 December 2022. Retrieved 22 January 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ↑ Wang K, Wei G, Liu D (November 2012). "CD19: a biomarker for B cell development, lymphoma diagnosis and therapy". Experimental Hematology & Oncology. 1 (1): 36. doi:10.1186/2162-3619-1-36. PMC 3520838. PMID 23210908.
- ↑ "Pyrrolobenzodiazepine". ADC Review. Archived from the original on 2 August 2017. Retrieved 2 August 2017.
- ↑ "Loncastuximab tesirine Orphan Drug Designations and Approvals". U.S. Food and Drug Administration (FDA). 8 June 2017. Archived from the original on 2 June 2021. Retrieved 1 June 2021.
- 1 2 "Zynlonta : Pending EC decision". European Medicines Agency (EMA). 15 September 2022. Archived from the original on 16 September 2022. Retrieved 18 September 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ "Zynlonta Product information". Union Register of medicinal products. Archived from the original on 10 January 2023. Retrieved 3 March 2023.
- ↑ Perrone S, Lopedote P, Levis M, Di Rocco A, Smith SD (March 2022). "Management of relapsed or refractory large B-cell lymphoma in patients ineligible for CAR-T cell therapy". Expert Review of Hematology. 15 (3): 215–232. doi:10.1080/17474086.2022.2044778. PMID 35184664. S2CID 247010986.
External links
- "Loncastuximab tesirine-lpyl". NCI Drug Dictionary. National Cancer Institute.
- "Loncastuximab tesirine-lpyl". National Cancer Institute. 21 May 2021.
- Clinical trial number NCT03589469 for "Study to Evaluate the Efficacy and Safety of Loncastuximab Tesirine in Patients With Relapsed or Refractory Diffuse Large B-Cell Lymphoma (LOTIS-2)" at ClinicalTrials.gov