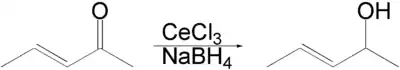Luche还原反应
Luche还原反应(Luche reduction)
α,β-不饱和酮在三氯化铈存在下为硼氢化钠选择性还原为相应的烯丙醇。[1][2][3]
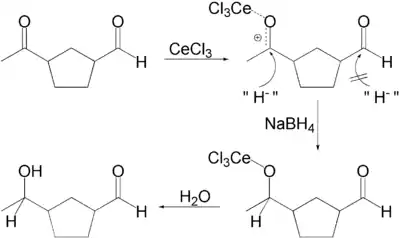
反应的选择性可通过软硬酸碱理论来解释。硼氢化钠发生甲醇解生成的甲氧基硼氢化钠是较硼氢化钠更“硬”的还原剂,因此倾向于与不饱和酮中较硬的1,2-羰基部分进行反应。而且,路易斯酸性的三氯化铈通过配位,增强了甲醇的活性,也提高了羰基的亲电性。
反應機構
参见
参考资料
- Strategic Applications of Named Reactions in Organic Synthesis (Paperback) by Laszlo Kurti, Barbara Czako ISBN 0-12-429785-4
- Lanthanides in organic chemistry. 1. Selective 1,2 reductions of conjugated ketones Jean Louis Luche J. Am. Chem. Soc.; 1978; 100(7); 2226-2227. doi:10.1021/ja00475a040
- Lanthanoids in organic synthesis. 6. Reduction of .alpha.-enones by sodium borohydride in the presence of lanthanoid chlorides: synthetic and mechanistic aspects Andre L. Gemal, Jean Louis Luche J. Am. Chem. Soc.; 1981; 103(18); 5454-5459 doi:10.1021/ja00408a029
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.