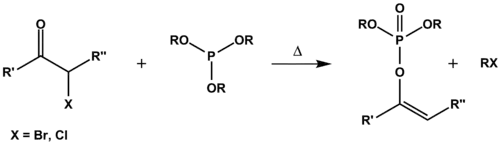
Perkow反应
反应机理
亚磷酸酯对卤代酮发生亲核取代,生成鏻盐,发生酮-烯醇互变异构,再受卤离子进攻,得最终产物磷酸酯。
如果不发生酮-烯醇异构,则得 Arbuzov 产物(以灰色表示)。
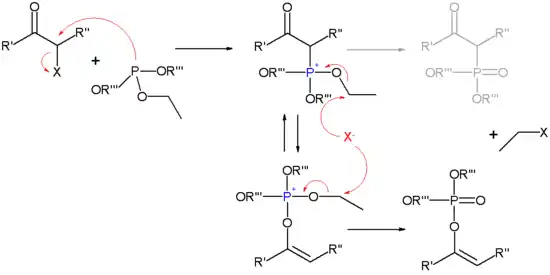
Perkow反应的机理。灰色显示的 Arbuzov 产物。
应用
1、六氯丙酮与亚磷酸三乙酯的 Perkow 产物,再用2,2,2-三氟乙醇钠处理得到的两性离子,与呋喃发生[4+3]环加成,用于新颖的驱虫剂之合成[2]
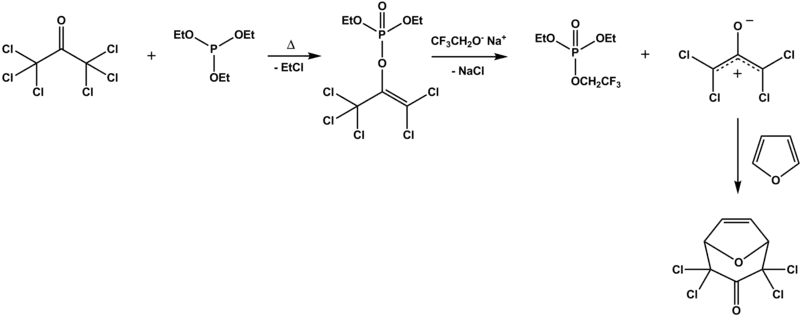
Perkow反应 例子
2、如下喹啉衍生物,取代基为正丁基时,亚磷酸酯加到C-4羰基上(作者推测),再经分子内重排,得到烯基磷酸酯;取代基为苯基时(未画出),亚磷酸酯加到三氟乙酰氧基的羰基上,经过重排后,最终得到乙基烯醇醚。[3]
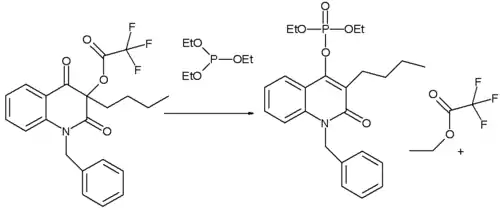
用于喹啉衍生物的合成。
参考资料
- Perkow, W. Chemische Berichte 1954, 87, 755–758
- Hexachloroacetone as a Precursor for a Tetrachloro-substituted Oxyallyl Intermediate: [4+3] Cycloaddition to Cyclic 1,3-Dienes Baldur Föhlisch and Stefan Reiner Molecules 2004, 9, 1-10 Online Article (页面存档备份,存于)
- New Modification of the Perkow Reaction: Halocarboxylate Anions as Leaving Groups in 3-Acyloxyquinoline-2,4(1H,3H)-dione Compounds Oldrich Paleta, Karel Pomeisl, Stanislav Kafka, Antonin Klasek, Vladislav Kubelka Beilstein Journal of Organic Chemistry 2005 Online Article (页面存档备份,存于)
- T. Moriguchi, K. Okada, K. Seio, and M. Sekine. "Synthesis and Stability of 1-Phenylethenyl Phosphate Derivatives and their Phosphoryl Transfer Activity", Letters in Organic Chemistry, 1 (2):140-144, 2004
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.