 | |
| Names | |
|---|---|
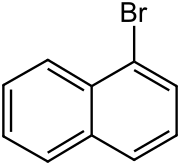
| Preferred IUPAC name
1-Bromonaphthalene | |
| Other names
α-Bromonaphthalene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.001.787 |
| EC Number |
|
| MeSH | C108222 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H7Br | |
| Molar mass | 207.070 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.48 g/mL |
| Melting point | 1–2 °C (34–36 °F; 274–275 K) |
| Boiling point | 132–135 °C at 12 mm; 145–148 °C at 20 mm |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
1-Bromonaphthalene is an organic compound with the formula C10H7Br.

It is one of two isomeric bromonaphthalenes, the other being 2-bromonaphthalene. Under normal conditions, the substance is a colorless liquid.
Synthesis and reactions
It is prepared by treatment of naphthalene with bromine:[1]
- C10H8 + Br2 → C10H7Br + HBr
The compound exhibits many reactions typical of aryl bromides. Bromide can be displaced by cyanide to give the nitrile. It forms a Grignard reagent[2] and organolithium compound. 1-Lithionaphthalene can be further lithiated to give 1,8-dilithionaphthalene, a precursor to peri-naphthalene compounds.
Applications
Because of its high refractive index (1.656-1.659nD),[3] 1-bromonaphthalene is used as an embedding agent in microscopy and for determining the refraction of crystals.
The compound is also used as a precursor to various substituted derivatives of naphthalene.
See also
References
- ↑ "α-BROMONAPHTHALENE". Organic Syntheses. 1: 35. 1921. doi:10.15227/orgsyn.001.0035.
- ↑ "α-NAPHTHOIC ACID". Organic Syntheses. 11: 80. 1931. doi:10.15227/orgsyn.011.0080.
- ↑ "1-Bromonaphthalene".