 | |
 | |
| Names | |
|---|---|
| IUPAC name
Aluminium nitrate | |
| Other names
Nitric Aluminum salt aluminum nitrate aluminium(III) nitrate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.033.396 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII |
|
| UN number | 1438 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
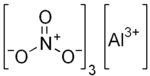
| Al(NO3)3 | |
| Molar mass | 212.996 g/mol (anhydrous) 375.134 g/mol (nonahydrate) |
| Appearance | White crystals, solid hygroscopic |
| Odor | odorless |
| Density | 1.72 g/cm3 (nonahydrate) |
| Melting point | 66 °C (151 °F; 339 K) (anhydrous)[1] 73.9 °C (165.0 °F; 347.0 K) (nonahydrate) |
| Boiling point | 150 °C (302 °F; 423 K) (nonahydrate) decomposes |
| anhydrous: 60.0 g/100ml (0°C) 73.9 g/100ml (20 °C) 160 g/100ml (100 °C) nonahydrate: 67.3 g/100 mL | |
| Solubility in methanol | 14.45 g/100ml |
| Solubility in ethanol | 8.63 g/100ml |
| Solubility in ethylene glycol | 18.32 g/100ml |
Refractive index (nD) |
1.54 |
| Hazards | |
| GHS labelling: | |
     | |
| Danger | |
| H271, H272, H301, H315, H318, H319, H361 | |
| P201, P202, P210, P220, P221, P264, P270, P280, P281, P283, P301+P310, P302+P352, P305+P351+P338, P306+P360, P308+P313, P310, P321, P330, P332+P313, P337+P313, P362, P370+P378, P371+P380+P375, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 35 °C (95 °F; 308 K) (nonahydrate) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
4280 mg/kg, oral (rat) |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
none[2] |
REL (Recommended) |
2 mg/m3[2] |
IDLH (Immediate danger) |
N.D.[2] |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Aluminium nitrate is a white, water-soluble salt of aluminium and nitric acid, most commonly existing as the crystalline hydrate, aluminium nitrate nonahydrate, Al(NO3)3·9H2O.
Preparation
Aluminium nitrate cannot be synthesized by the reaction of aluminium with concentrated nitric acid, as the aluminium forms a passivation layer.
Aluminium nitrate may instead be prepared by the reaction of nitric acid with aluminium(III) chloride. Nitrosyl chloride is produced as a by-product; it bubbles out of the solution as a gas. More conveniently, the salt can be made by reacting nitric acid with aluminium hydroxide.
Aluminium nitrate may also be prepared a metathesis reaction between aluminium sulfate and a nitrate salt with a suitable cation such as barium, strontium, calcium, silver, or lead. e.g. Al2(SO4)3 + 3 Ba(NO3)2 → 2 Al(NO3)3 + 3 BaSO4.
Uses
Aluminium nitrate is a strong oxidizing agent. It is used in tanning leather, antiperspirants, corrosion inhibitors, extraction of uranium, petroleum refining, and as a nitrating agent.
The nonahydrate and other hydrated aluminium nitrates have many applications. These salts are used to produce alumina for preparation of insulating papers, in cathode ray tube heating elements, and on transformer core laminates. The hydrated salts are also used for the extraction of actinide elements.[3]
It is used in the laboratory and classroom such as in the reaction
- Al(NO3)3 + 3 NaOH → Al(OH)3 + 3 NaNO3
It is, however, much less often encountered than aluminium chloride and aluminium sulfate.
References
- ↑ "aluminum nitrate".
- 1 2 3 NIOSH Pocket Guide to Chemical Hazards. "#0024". National Institute for Occupational Safety and Health (NIOSH).
- ↑ Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8.
