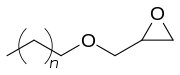
 n = 10-12 | |
| Names | |
|---|---|
| IUPAC name
2-(dodecoxymethyl)oxirane;2-(tetradecoxymethyl)oxirane;2-(tridecoxymethyl)oxirane | |
| Other names
Alkyl glycidyl ether | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.065.295 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C48H96O6 | |
| Molar mass | 769.3 g/mol |
| Hazards | |
| GHS labelling:[1] | |
 | |
| Warning | |
| H315, H317 | |
| P261, P264, P272, P280, P302+P352, P321, P332+P313, P333+P313, P362, P363, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
C12-C14 alcohol glycidyl ether (AGE) is an organic chemical in the glycidyl ether family.[2] It is a mixture of mainly 12 and 14 carbon chain alcohols, also called fatty alcohols that have been glycidated. It is an industrial chemical used as a surfactant but primarily for epoxy resin viscosity reduction.[3] It has the CAS number 68609-97-2 but the IUPAC name is more complex as it is a mixture and is 2-(dodecoxymethyl)oxirane;2-(tetradecoxymethyl)oxirane;2-(tridecoxymethyl)oxirane.[4] Other names include dodecyl and tetradecyl glycidyl ethers and alkyl (C12-C14) glycidyl ether.
Manufacture
A fatty alcohol mixture rich in C12-C14 alcohols is placed in a reactor with a Lewis acid catalyst. Then epichlorohydrin is added slowly to control exotherm which results in the formation of the halohydrins.[5] This is followed by a caustic dehydrochlorination, to form C12-C14 alcohol glycidyl ether.[6] The waste products are water and sodium chloride and excess caustic soda.[7] One of the quality control tests would involve measuring the Epoxy value by determination of the epoxy equivalent weight.
Uses
As an epoxy modifier it is classed as an epoxy reactive diluent.[8] It is one of a family of glycidyl ethers available used for viscosity reduction of epoxy resins.[9] These are then further formulated into coatings, sealants, adhesives, and elastomers.[10][11] Resins with this diluent tend to show improved workability.[12] It is also used to synthesize other molecules.[13][14] The use of the diluent does effect mechanical properties and microstructure of epoxy resins.[15][16]
Toxicology
The toxicology is well known, and it is classed as a skin irritant.[17]
See also
References
- ↑ "Dodecyl and tetradecyl glycidyl ethers". pubchem.ncbi.nlm.nih.gov. Retrieved 12 April 2022.
- ↑ "Alkyl (C12-C14) glycidyl ether | 68609-97-2". www.chemicalbook.com. Retrieved 2022-04-10.
- ↑ Jagtap, Ameya Rajendra; More, Aarti (2022-08-01). "Developments in reactive diluents: a review". Polymer Bulletin. 79 (8): 5667–5708. doi:10.1007/s00289-021-03808-5. ISSN 1436-2449. S2CID 235678040.
- ↑ PubChem. "Dodecyl and tetradecyl glycidyl ethers". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-04-12.
- ↑ "Process for the preparation of glycidyl ethers- US Patent 5162547" (PDF). November 1992.
- ↑ SJÖVOLD, HENRICK (2015). "Solvent-Free Synthesis of Glycidyl Ethers : Investigating Factors Influencing the Yield of Alkyl Glycidyl Ethers Master of Science Thesis" (PDF). Chalmers University Sweden.
- ↑ "Preparation method of alkyl glycidyl ether - Patent CN-113429367-A - PubChem". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-04-12.
- ↑ Monte, Salvatore J. (1998), Pritchard, Geoffrey (ed.), "Diluents and viscosity modifiers for epoxy resins", Plastics Additives: An A-Z reference, Polymer Science and Technology Series, Dordrecht: Springer Netherlands, vol. 1, pp. 211–216, doi:10.1007/978-94-011-5862-6_24, ISBN 978-94-011-5862-6, retrieved 2022-03-29
- ↑ Ali, M.; Hammami, A. (July 2005). "Experimental modeling of the cure behavior of a formulated blend of DGEBA epoxy and C12-C14 glycidyl ether as a reactive diluent". Polymer Composites. 26 (5): 593–603. doi:10.1002/pc.20131. ISSN 0272-8397.
- ↑ Howarth G.A "Synthesis of a legislation compliant corrosion protection coating system based on urethane, oxazolidine and waterborne epoxy technology" pages 23,24,39 Master of Science Thesis April 1997 Imperial College London
- ↑ Monte, Salvatore J. (1998), Pritchard, Geoffrey (ed.), "Diluents and viscosity modifiers for epoxy resins", Plastics Additives: An A-Z reference, Polymer Science and Technology Series, Dordrecht: Springer Netherlands, vol. 1, pp. 211–216, doi:10.1007/978-94-011-5862-6_24, ISBN 978-94-011-5862-6, retrieved 2022-04-12
- ↑ Ozeren Ozgul, Eren; Ozkul, M. Hulusi (2018-01-15). "Effects of epoxy, hardener, and diluent types on the workability of epoxy mixtures". Construction and Building Materials. 158: 369–377. doi:10.1016/j.conbuildmat.2017.10.008. ISSN 0950-0618.
- ↑ Urata, Kouichi; Takaishi, Naotake (September 1994). "The alkyl glycidyl ether as synthetic building blocks". Journal of the American Oil Chemists' Society. 71 (9): 1027–1033. doi:10.1007/BF02542274. S2CID 96776835.
- ↑ Verkoyen, Patrick; Frey, Holger (August 2020). "Long‐Chain Alkyl Epoxides and Glycidyl Ethers: An Underrated Class of Monomers". Macromolecular Rapid Communications. 41 (15): 2000225. doi:10.1002/marc.202000225. ISSN 1022-1336. PMID 32567153.
- ↑ Khalina, Morteza; Beheshty, Mohammad Hosain; Salimi, Ali (2019-08-01). "The effect of reactive diluent on mechanical properties and microstructure of epoxy resins". Polymer Bulletin. 76 (8): 3905–3927. doi:10.1007/s00289-018-2577-6. ISSN 1436-2449. S2CID 105389177.
- ↑ Pastarnokienė, Liepa; Jonikaitė-Švėgždienė, Jūratė; Lapinskaitė, Neringa; Kulbokaitė, Rūta; Bočkuvienė, Alma; Kochanė, Tatjana; Makuška, Ričardas (2023-07-01). "The effect of reactive diluents on curing of epoxy resins and properties of the cured epoxy coatings". Journal of Coatings Technology and Research. 20 (4): 1207–1221. doi:10.1007/s11998-022-00737-4. ISSN 1935-3804. S2CID 256749849.
- ↑ "C&L Inventory". echa.europa.eu. Retrieved 2022-04-12.
Further reading
- Epoxy resin technology. Paul F. Bruins, Polytechnic Institute of Brooklyn. New York: Interscience Publishers. 1968. ISBN 0-470-11390-1. OCLC 182890.
{{cite book}}: CS1 maint: others (link) - Flick, Ernest W. (1993). Epoxy resins, curing agents, compounds, and modifiers : an industrial guide. Park Ridge, NJ. ISBN 978-0-8155-1708-5. OCLC 915134542.
{{cite book}}: CS1 maint: location missing publisher (link) - Lee, Henry (1967). Handbook of epoxy resins. Kris Neville ([2nd, expanded work] ed.). New York: McGraw-Hill. ISBN 0-07-036997-6. OCLC 311631322.
- "Dow Epoxy Resins" (PDF).