| CLPB | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | CLPB, HSP78, SKD3, ANKCLB, MEGCANN, MGCA7, ClpB homolog, mitochondrial AAA ATPase chaperonin, caseinolytic mitochondrial matrix peptidase chaperone subunit B, SCN9, MGCA7A | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 616254 MGI: 1100517 HomoloGene: 32067 GeneCards: CLPB | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
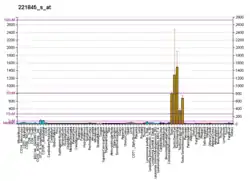
Caseinolytic peptidase B protein homolog (CLPB), also known as Skd3, is a mitochondrial AAA ATPase chaperone that in humans is encoded by the gene CLPB,[5][6][7] which encodes an adenosine triphosphate-(ATP) dependent chaperone. Skd3 is localized in mitochondria and widely expressed in human tissues. High expression in adult brain and low expression in granulocyte is found.[8][9] It is a potent protein disaggregase that chaperones the mitochondrial intermembrane space.[10] Mutations in the CLPB gene could cause autosomal recessive metabolic disorder with intellectual disability/developmental delay, congenital neutropenia, progressive brain atrophy, movement disorder, cataracts, and 3-methylglutaconic aciduria.[8][11] Recently, heterozygous, dominant negative mutations in CLPB have been identified as a cause of severe congenital neutropenia (SCN).[12]
Structure
Gene
The CLPB gene has 19 exons and is located at the chromosome band 11q13.4.[7]
Protein
Skd3 has five isoforms due to alternative splicing. Isoform 1 is considered to have the 'canonical' sequence. The protein is 78.7 kDa in size and composed of 707 amino acids. It contains an N-terminal mitochondrial targeting sequence (1-92 amino acids).[10] After processing, the mature mitochondrial protein has a theoretical pI of 7.53.[13] Skd3 is further processed by the mitochondrial rhomboid protease PARL at amino acid 127.[10][14] Skd3 has a specific C-terminal D2 domain and proteins with this domain form the sub-family of Caseinolytic peptidase (Clp) proteins, also called HSP100.[15] The domain composition of human Skd3 is different from that of microbial or plant orthologs.[10][16] Notably, the presence of ankyrin repeats replaced the first of two ATPase domains found in bacteria and fungi.[17][18]
Function
Skd3 belongs to the HCLR clade of the large AAA+ superfamily.[10][19] The unifying characteristic of this family is the hydrolysis of ATP through the AAA+ domain to produce energy required to catalyze protein unfolding, disassembly and disaggregation.[20][21] Skd3 does not cooperate with HSP70, unlike its bacterial orthologue.[10] The in vitro ATPase activity of Skd3 has been confirmed.[8][10][22] Skd3 is a potent disaggregase in vitro and is activated by PARL to increase disaggregation activity by over 10-fold.[10] Indeed, PARL-activated Skd3 is capable of disassembling alpha-synuclein fibrils in vitro.[10] Even though the bacterial orthologue, ClpB, contributes to the thermotolerance of cells, it is yet unclear if Skd3 plays a similar role within mitochondria.[20][23] The interaction with protein like HAX1 suggests that human Skd3 may be involved in apoptosis.[8] Indeed, Skd3 solubilizes HAX1 in cells and the deletion of the CLPB gene in human cells has been shown to sensitize cells to apoptotic signals.[10][24] In humans, the presence of ankyrin repeats replaced the first of two ATPase domains found in bacteria and fungi, which might have evolved to ensure more elaborate substrate recognition or to support a putative chaperone function.[17][18] Either the ankyrin repeats alone or the AAA+ domain were found to be insufficient to support disaggregation activity.[10] With only one ATPase domain, Skd3 is postulated competent in the use of ATP hydrolysis energy for threading unfolded polypeptide through the central channel of the hexamer ring.[25][26][27] />
Clinical significance
Neonatal encephalopathy is a kind of severe neurological impairment in the newborn with no specific clinical sign at the early stage of life, and its diagnosis remains a challenge. This neonatal encephalopathy includes a heterogeneous group of 3-methylglutaconic aciduria syndromes and loss of Skd3 function is reported to be one of the causes. Knocking down the clpB gene in the zebrafish induced reduction of growth and increment of motor activity, which is similar to the signs observed in patients.[20] Its loss may lead to a broad phenotypic spectrum encompassing intellectual disability/developmental delay, congenital neutropenia, progressive brain atrophy, movement disorder, and bilateral cataracts, with 3-methylglutaconic aciduria.[8][11][28] Further investigation into Skd3 may shed a new light on the diagnosis of this disease.
Interactions
This protein is known to interact with:
References
- 1 2 3 GRCh38: Ensembl release 89: ENSG00000162129 - Ensembl, May 2017
- 1 2 3 GRCm38: Ensembl release 89: ENSMUSG00000001829 - Ensembl, May 2017
- ↑ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ Wiemann S, Weil B, Wellenreuther R, Gassenhuber J, Glassl S, Ansorge W, Böcher M, Blöcker H, Bauersachs S, Blum H, Lauber J, Düsterhöft A, Beyer A, Köhrer K, Strack N, Mewes HW, Ottenwälder B, Obermaier B, Tampe J, Heubner D, Wambutt R, Korn B, Klein M, Poustka A (March 2001). "Toward a catalog of human genes and proteins: sequencing and analysis of 500 novel complete protein coding human cDNAs". Genome Research. 11 (3): 422–35. doi:10.1101/gr.GR1547R. PMC 311072. PMID 11230166.
- ↑ Périer F, Radeke CM, Raab-Graham KF, Vandenberg CA (January 1995). "Expression of a putative ATPase suppresses the growth defect of a yeast potassium transport mutant: identification of a mammalian member of the Clp/HSP104 family". Gene. 152 (2): 157–63. doi:10.1016/0378-1119(94)00697-Q. PMID 7835694.
- 1 2 "Entrez Gene: CLPB ClpB caseinolytic peptidase B homolog (E. coli)".
- 1 2 3 4 5 6 Wortmann SB, Ziętkiewicz S, Kousi M, Szklarczyk R, Haack TB, Gersting SW, et al. (February 2015). "CLPB mutations cause 3-methylglutaconic aciduria, progressive brain atrophy, intellectual disability, congenital neutropenia, cataracts, movement disorder". American Journal of Human Genetics. 96 (2): 245–57. doi:10.1016/j.ajhg.2014.12.013. PMC 4320260. PMID 25597510.
- ↑ Saunders C, Smith L, Wibrand F, Ravn K, Bross P, Thiffault I, Christensen M, Atherton A, Farrow E, Miller N, Kingsmore SF, Ostergaard E (February 2015). "CLPB variants associated with autosomal-recessive mitochondrial disorder with cataract, neutropenia, epilepsy, and methylglutaconic aciduria". American Journal of Human Genetics. 96 (2): 258–65. doi:10.1016/j.ajhg.2014.12.020. PMC 4320254. PMID 25597511.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 Cupo, Ryan R; Shorter, James (2020-06-23). Berger, James M (ed.). "Skd3 (human CLPB) is a potent mitochondrial protein disaggregase that is inactivated by 3-methylglutaconic aciduria-linked mutations". eLife. 9: e55279. doi:10.7554/eLife.55279. ISSN 2050-084X. PMC 7343390. PMID 32573439.
- 1 2 Kiykim A, Garncarz W, Karakoc-Aydiner E, Ozen A, Kiykim E, Yesil G, Boztug K, Baris S (April 2016). "Novel CLPB mutation in a patient with 3-methylglutaconic aciduria causing severe neurological involvement and congenital neutropenia". Clinical Immunology. 165: 1–3. doi:10.1016/j.clim.2016.02.008. PMID 26916670.
- ↑ Warren, Julia T; Cupo, Ryan R; Wattanasirakul, Peeradol; Spencer, David; Locke, Adam E; Makaryan, Vahagn; Bolyard, Audrey Anna; Kelley, Meredith L; Kingston, Natalie L; Shorter, James; Bellanné-Chantelot, Christine (2021-06-11). "Heterozygous Variants of CLPB are a Cause of Severe Congenital Neutropenia". Blood. 139 (blood.2021010762): 779–791. doi:10.1182/blood.2021010762. ISSN 0006-4971. PMC 8814677. PMID 34115842.
- ↑ "Q9H078 - CLPB_HUMAN". Uniprot.
- 1 2 Saita, Shotaro; Nolte, Hendrik; Fiedler, Kai Uwe; Kashkar, Hamid; Venne, A. Saskia; Zahedi, René P.; Krüger, Marcus; Langer, Thomas (April 2017). "PARL mediates Smac proteolytic maturation in mitochondria to promote apoptosis". Nature Cell Biology. 19 (4): 318–328. doi:10.1038/ncb3488. ISSN 1476-4679. PMID 28288130. S2CID 3744933.
- ↑ Zolkiewski M (September 2006). "A camel passes through the eye of a needle: protein unfolding activity of Clp ATPases". Molecular Microbiology. 61 (5): 1094–100. doi:10.1111/j.1365-2958.2006.05309.x. PMC 1852505. PMID 16879409.
- ↑ Erives, Albert J.; Fassler, Jan S. (2015-02-24). "Metabolic and Chaperone Gene Loss Marks the Origin of Animals: Evidence for Hsp104 and Hsp78 Chaperones Sharing Mitochondrial Enzymes as Clients". PLOS ONE. 10 (2): e0117192. Bibcode:2015PLoSO..1017192E. doi:10.1371/journal.pone.0117192. ISSN 1932-6203. PMC 4339202. PMID 25710177.
- 1 2 Mosavi LK, Cammett TJ, Desrosiers DC, Peng ZY (June 2004). "The ankyrin repeat as molecular architecture for protein recognition". Protein Science. 13 (6): 1435–48. doi:10.1110/ps.03554604. PMC 2279977. PMID 15152081.
- 1 2 Li J, Mahajan A, Tsai MD (December 2006). "Ankyrin repeat: a unique motif mediating protein-protein interactions". Biochemistry. 45 (51): 15168–78. CiteSeerX 10.1.1.502.2771. doi:10.1021/bi062188q. PMID 17176038.
- ↑ Erzberger, Jan P.; Berger, James M. (2006-05-11). "Evolutionary relationships and structural mechanisms of aaa+ proteins". Annual Review of Biophysics and Biomolecular Structure. 35 (1): 93–114. doi:10.1146/annurev.biophys.35.040405.101933. ISSN 1056-8700. PMID 16689629.
- 1 2 3 Capo-Chichi JM, Boissel S, Brustein E, Pickles S, Fallet-Bianco C, Nassif C, Patry L, Dobrzeniecka S, Liao M, Labuda D, Samuels ME, Hamdan FF, Vande Velde C, Rouleau GA, Drapeau P, Michaud JL (May 2015). "Disruption of CLPB is associated with congenital microcephaly, severe encephalopathy and 3-methylglutaconic aciduria". Journal of Medical Genetics. 52 (5): 303–11. doi:10.1136/jmedgenet-2014-102952. PMID 25650066. S2CID 36062854.
- ↑ Snider J, Thibault G, Houry WA (30 April 2008). "The AAA+ superfamily of functionally diverse proteins". Genome Biology. 9 (4): 216. doi:10.1186/gb-2008-9-4-216. PMC 2643927. PMID 18466635.
- ↑ Mróz, Dagmara; Wyszkowski, Hubert; Szablewski, Tomasz; Zawieracz, Katarzyna; Dutkiewicz, Rafał; Bury, Katarzyna; Wortmann, Saskia B.; Wevers, Ron A.; Ziętkiewicz, Szymon (April 2020). "CLPB (caseinolytic peptidase B homolog), the first mitochondrial protein refoldase associated with human disease". Biochimica et Biophysica Acta (BBA) - General Subjects. 1864 (4): 129512. doi:10.1016/j.bbagen.2020.129512. hdl:2066/219662. PMID 31917998.
- ↑ Thomas JG, Baneyx F (October 1998). "Roles of the Escherichia coli small heat shock proteins IbpA and IbpB in thermal stress management: comparison with ClpA, ClpB, and HtpG In vivo". Journal of Bacteriology. 180 (19): 5165–72. doi:10.1128/JB.180.19.5165-5172.1998. PMC 107554. PMID 9748451.
- 1 2 3 Chen, Xufeng; Glytsou, Christina; Zhou, Hua; Narang, Sonali; Reyna, Denis E.; Lopez, Andrea; Sakellaropoulos, Theodore; Gong, Yixiao; Kloetgen, Andreas; Yap, Yoon Sing; Wang, Eric (July 2019). "Targeting Mitochondrial Structure Sensitizes Acute Myeloid Leukemia to Venetoclax Treatment". Cancer Discovery. 9 (7): 890–909. doi:10.1158/2159-8290.CD-19-0117. ISSN 2159-8274. PMC 6606342. PMID 31048321.
- ↑ Horwich AL (November 2004). "Chaperoned protein disaggregation--the ClpB ring uses its central channel". Cell. 119 (5): 579–81. doi:10.1016/j.cell.2004.11.018. PMID 15550237.
- ↑ Weibezahn J, Tessarz P, Schlieker C, Zahn R, Maglica Z, Lee S, Zentgraf H, Weber-Ban EU, Dougan DA, Tsai FT, Mogk A, Bukau B (November 2004). "Thermotolerance requires refolding of aggregated proteins by substrate translocation through the central pore of ClpB". Cell. 119 (5): 653–65. doi:10.1016/j.cell.2004.11.027. PMID 15550247.
- ↑ Nakazaki Y, Watanabe YH (December 2014). "ClpB chaperone passively threads soluble denatured proteins through its central pore". Genes to Cells. 19 (12): 891–900. doi:10.1111/gtc.12188. PMID 25288401. S2CID 7170147.
- ↑ Pronicka E, Piekutowska-Abramczuk D, Ciara E, Trubicka J, Rokicki D, Karkucińska-Więckowska A, Pajdowska M, Jurkiewicz E, Halat P, Kosińska J, Pollak A, Rydzanicz M, Stawinski P, Pronicki M, Krajewska-Walasek M, Płoski R (12 June 2016). "New perspective in diagnostics of mitochondrial disorders: two years' experience with whole-exome sequencing at a national paediatric centre". Journal of Translational Medicine. 14 (1): 174. doi:10.1186/s12967-016-0930-9. PMC 4903158. PMID 27290639.
- ↑ Yoshinaka, Takahiro; Kosako, Hidetaka; Yoshizumi, Takuma; Furukawa, Ryo; Hirano, Yu; Kuge, Osamu; Tamada, Taro; Koshiba, Takumi (2019-09-27). "Structural Basis of Mitochondrial Scaffolds by Prohibitin Complexes: Insight into a Role of the Coiled-Coil Region". iScience. 19: 1065–1078. Bibcode:2019iSci...19.1065Y. doi:10.1016/j.isci.2019.08.056. ISSN 2589-0042. PMC 6745515. PMID 31522117.
External links
- Human clpB genome location and clpB gene details page in the UCSC Genome Browser.
Further reading
- Rual JF, Venkatesan K, Hao T, Hirozane-Kishikawa T, Dricot A, Li N, Berriz GF, Gibbons FD, Dreze M, Ayivi-Guedehoussou N, Klitgord N, Simon C, Boxem M, Milstein S, Rosenberg J, Goldberg DS, Zhang LV, Wong SL, Franklin G, Li S, Albala JS, Lim J, Fraughton C, Llamosas E, Cevik S, Bex C, Lamesch P, Sikorski RS, Vandenhaute J, Zoghbi HY, Smolyar A, Bosak S, Sequerra R, Doucette-Stamm L, Cusick ME, Hill DE, Roth FP, Vidal M (October 2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature. 437 (7062): 1173–8. Bibcode:2005Natur.437.1173R. doi:10.1038/nature04209. PMID 16189514. S2CID 4427026.
- Colland F, Jacq X, Trouplin V, Mougin C, Groizeleau C, Hamburger A, Meil A, Wojcik J, Legrain P, Gauthier JM (July 2004). "Functional proteomics mapping of a human signaling pathway". Genome Research. 14 (7): 1324–32. doi:10.1101/gr.2334104. PMC 442148. PMID 15231748.
- Leonard D, Ajuh P, Lamond AI, Legerski RJ (September 2003). "hLodestar/HuF2 interacts with CDC5L and is involved in pre-mRNA splicing". Biochemical and Biophysical Research Communications. 308 (4): 793–801. CiteSeerX 10.1.1.539.8359. doi:10.1016/S0006-291X(03)01486-4. PMID 12927788.