 | |
| Names | |
|---|---|
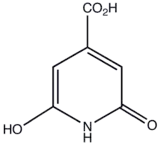
| Preferred IUPAC name
6-Hydroxy-2-oxo-1,2-dihydropyridine-4-carboxylic acid | |
| Other names
2,6-Dihydroxyisonicotinic acid; 2,6-Dihydroxypyridine-4-carboxylic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.002.483 |
| EC Number |
|
| MeSH | acid Citrazinic acid |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H5NO4 | |
| Molar mass | 155.109 g·mol−1 |
| Appearance | yellow powder |
| Melting point | >300 °C (decomp.) |
| Slightly soluble | |
| Solubility | Slightly soluble in methanol, DMSO, acidic solutions Very soluble in alkaline solutions |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P280, P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
> 3200 mg/kg ( Rat ) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Citrazinic acid (CZA) is a heterocyclic compound consisting of a dihydropyridine ring derived with a carboxylate group. The yellow solid exists as multiple tautomers, and it frequently forms dimers.[1]
Citrazinic acid is commonly formed in citric acid based carbon nanodots (CND). It is responsible for the blue light found in citric acid CNDs. The wavelengths of light emitted by citrazinic in CNDs can be shifted by changing the pH of the solution.[2]
Preparation
Citrazinic acid can be prepared creating a solution of citric acid and toluenesulfonic acid, which forms a 1,3-diester. That solution, when added to a heated ammonia solution results in citrazinic acid.[3]
References
- ↑ Stagi L, Mura S, Malfatti L, Carbonaro CM, Ricci PC, Porcu S, et al. (May 2020). "Anomalous Optical Properties of Citrazinic Acid under Extreme pH Conditions". ACS Omega. 5 (19): 10958–10964. doi:10.1021/acsomega.0c00775. PMC 7241015. PMID 32455216.
- ↑ Mocci F, Olla C, Cappai A, Corpino R, Ricci PC, Chiriu D, et al. (February 2021). "Formation of Citrazinic Acid Ions and Their Contribution to Optical and Magnetic Features of Carbon Nanodots: A Combined Experimental and Computational Approach". Materials. 14 (4): 770. Bibcode:2021Mate...14..770M. doi:10.3390/ma14040770. PMC 7914458. PMID 33562081.
- ↑ Baizer MM, Dub M, Gister S, Steinberg NG (July 1956). "Synthesis of isoniazid from citric acid". Journal of the American Pharmaceutical Association. 45 (7): 478–480. doi:10.1002/jps.3030450714. PMID 13345683.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.
