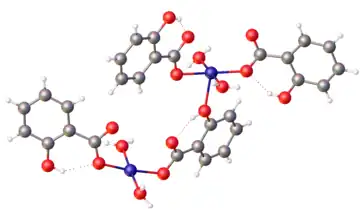
Structure of a hydrated copper(II) salicylate.[1] Color code: red = O, blue = Cu, gray = C, white = H.
Copper salicylate describes a range of compounds containing copper(II) and salicylate. Many compounds are known. They are generally blue. Simple species include:
- Cu2(2O2C6H4OH)4(H2O)2·L) (L = diverse solvent.[4]
Many copper(II) salicylates are known with amines and N-hetrerocyclic ligands. None appear to have commercial applications.
References
- ↑ Lutz, Martin; Kroon-Batenburg, Loes M. J. (2018). "Order-Disorder in Diaquobis(salicylato)copper(II) Revisited". Croatica Chemica Acta. 91 (2). doi:10.5562/cca3362. S2CID 59025247.
- ↑ Jagner, S.; Hazell, R. G.; Larsen, K. P. (1976). "The crystal structure of diaquabis(salicylato)copper(II), Cu[C6H4(OH)COO]2(H2O)2". Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry. 32 (2): 548–554. Bibcode:1976AcCrB..32..548J. doi:10.1107/S0567740876003397.
- ↑ Hanic, F.; Michalov, J. (1960). "Die Kristallstruktur von Kupfersalicylat-Tetrahydrat Cu(OH.C6H4.COO)2.4H2O". Acta Crystallographica. 13 (4): 299–302. Bibcode:1960AcCry..13..299H. doi:10.1107/S0365110X60000753.
- ↑ V.V.Gorinchoy, Yu.A.Simonov, S.G.Shova, Y.N.Szafranski, K.I.Turta (2009). Zh.Strukt.Khim.(Russ.)(J.Struct.Chem.). 50: 1196.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.