 | |
 | |
| Names | |
|---|---|
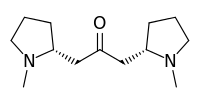
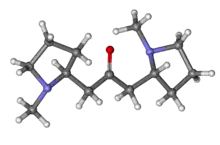
| Preferred IUPAC name
1-[(2R)-1-Methylpyrrolidin-2-yl]-3-[(2S)-1-methylpyrrolidin-2-yl]propan-2-one | |
| Identifiers | |
3D model (JSmol) |
|
| 3DMet | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C13H24N2O | |
| Molar mass | 224.348 g·mol−1 |
| Melting point | 40–41 °C (104–106 °F; 313–314 K) (trihydrate) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Cuscohygrine is a pyrrolidine alkaloid found in coca. It can also be extracted from plants of the family Solanaceae, including Atropa belladonna (deadly nightshade), Datura innoxia and Datura stramonium (jimson weed). Cuscohygrine usually occurs along with other, more potent alkaloids such as atropine or cocaine.
Cuscohygrine, along with the related metabolite hygrine, was first isolated by Carl Liebermann in 1889 as an alkaloid accompanying cocaine in coca leaves (also known as Cusco-leaves).
Cuscohygrine is an oil that can be distilled without decomposition only in vacuum. It is soluble in water. It also forms a crystalline trihydrate which melts at 40–41 °C.
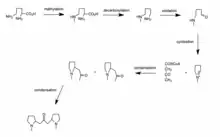
Biosynthesis
Ornithine is methylated to N-methylornithine and when decarboxylated, becomes N-methylputrescine. 4-methylaminobutanal is yielded from the oxidation of the primary amino-group. 4-methylaminobutanal then cyclizes to an N-methyl-l-pyrrolinium salt. The condensation of the pyrrolinium salt with acetoacetyl coenzyme A yields hygrine. Finally, the condensation of the hygrine molecule with another molecule of pyrrolidinium salt yields cuscohygrine.[1]
See also
References
- ↑ O'Donovan, D. G.; Keogh, M. F. (1969). "The role of hygrine in the biosynthesis of cuscohygrine and hyoscyamine". Journal of the Chemical Society C: Organic (2): 223–226. doi:10.1039/J39690000223.
- "USDA, ARS, National Genetic Resources Program. Phytochemical and Ethnobotanical Databases.[Online Database] National Germplasm Resources Laboratory, Beltsville, Maryland". Archived from the original on December 12, 2012. Retrieved July 14, 2005.
- Dr. Ame Pictet (1904). The Vegetable Alkaloids. With particular reference to their chemical constitution. London: Chapman & Hall.