 | |
| Names | |
|---|---|
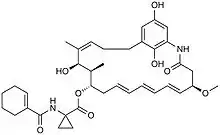
| IUPAC name
[(5R,6E,8E,10E,13S,14R,15R,16Z)-15,22,24-trihydroxy-5-methoxy-14,16-dimethyl-3-oxo-2-azabicyclo[18.3.1]tetracosa-1(23),6,8,10,16,20(24),21-heptaen-13-yl] 1-(cyclohexene-1-carbonylamino)cyclopropane-1-carboxylate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| MeSH | C106761 |
PubChem CID |
|
| |
| |
| Properties | |
| C37H48N2O8 | |
| Molar mass | 648.797 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Cytotrienin A (Cyt A) is a secondary metabolite isolated from Streptomyces sp. RK95-74 isolated from soil in Japan in 1997.[1][2] Cyt A is an ansamycin.[2] Cytotrienin A induces apoptosis on HL-60 cells, as well as inhibiting translation in eukaryotes by inhibiting eukaryotic elongation factor 1A (eEF1A), which can act as an oncogene.[1][3] These functions lead to the potential of the microbial metabolite acting as an anticancer agent, specifically for blood cancers, as it has proved to be more effective with leukemic cell lines.[1][3] Cyt A is thought to induce apoptosis by activating c-Jun N-terminal Kinase (JNK), p38 mitogen-activated protein kinase (MAPK), and p36 myelin basic protein (MBP) kinase.[3][4]
Structure
Cytotrienin A is an ansamycin with a macrocyclic, twenty-one carbon, lactam, featuring an E,E,E-triene and an unusual aminocyclopropane carboxylic acid side chain.[2] The structure features four stereocenters, 3 of which are contiguous. The absolute stereochemistry of the naturally occurring metabolite revealed it to be (+)-Cytotrienin A.[2]
Biology and Biochemistry
Cytotrienin A has a median effective dose value of 7.7 nM to induce apoptosis on HL-60 cells.[1] It was also shown that the metabolite has greater growth inhibitory activity on HL-60 cells at low concentrations and little impact on non-tumorous cells, while at high concentrations the reverse relationship was seen.[1] The apoptosis pathway was shown to involve the proteolytic activation of MST/Krs proteins by caspase-3 which results in the activation of p36 MBP kinase through the creation of ROS. The concentrations needed to induce this pathway were found to be the same required to induce apoptosis on HL-60 cells.[4][5] Cyt A also activates c-Jun N-terminal kinase, and its apoptotic effects are inhibited by dominant negative c-Jun. While the presence of inactive MST/Krs proteins and dominant negative c-Jun completely inhibited apoptosis.[4]
Additionally, cytotrienin A acts to inhibit eEF-1A, and thus inhibiting translation. This can make the tumor less effective at producing anti-apoptotic mediators and lowering the tumor's drug resistance.[3] The translation inhibition is thought to occur before apoptosis can be detected. Inhibition of HUVEC tube formation was also shown, which indicates an inhibition of angiogenesis and further evidence of its anti-cancer potential.[3] In A549 cells, where cytotrienin A has been shown to be less effective, cyt A inhibits the expression of ICAM-1. This occurs when the TNF receptor 1 is shredded through the activation of extracellular signal-regulated kinase (ERK) and p38 MAPK.[3][6] Cyt A inhibited ICAM-1 expression by TNF-α and IL-1α at similar concentrations, but was found to be inhibited by TAPI-2, an inhibitor to the TNF-α-converting enzyme (TACE).[6]
References
- 1 2 3 4 5 Kakeya, H.; Zhang, H. P.; Kobinata, K.; Onose, R.; Onozawa, C.; Kudo, T.; Osada, H. (April 1997). "Cytotrienin A, a novel apoptosis inducer in human leukemia HL-60 cells". The Journal of Antibiotics. 50 (4): 370–372. doi:10.7164/antibiotics.50.370. ISSN 0021-8820. PMID 9186568.
- 1 2 3 4 Hayashi, Yujiro; Shoji, Mitsuru; Ishikawa, Hayato; Yamaguchi, Junichiro; Tamura, Tomohiro; Imai, Hiroki; Nishigaya, Yosuke; Takabe, Kenichi; Kakeya, Hideaki; Osada, Hiroyuki (2008-08-18). "The Asymmetric Total Synthesis of (+)‐Cytotrienin A, an Ansamycin‐Type Anticancer Drug". Angewandte Chemie International Edition. 47 (35): 6657–6660. doi:10.1002/anie.200802079. ISSN 1433-7851. PMID 18646025.
- 1 2 3 4 5 6 Lindqvist, Lisa; Robert, Francis; Merrick, William; Kakeya, Hideaki; Fraser, Christopher; Osada, Hiroyuki; Pelletier, Jerry (December 2010). "Inhibition of translation by cytotrienin A--a member of the ansamycin family". RNA (New York, N.Y.). 16 (12): 2404–2413. doi:10.1261/rna.2307710. ISSN 1469-9001. PMC 2995401. PMID 20943818.
- 1 2 3 Watabe, M.; Kakeya, H.; Onose, R.; Osada, H. (2000-03-24). "Activation of MST/Krs and c-Jun N-terminal kinases by different signaling pathways during cytotrienin A-induced apoptosis". The Journal of Biological Chemistry. 275 (12): 8766–8771. doi:10.1074/jbc.275.12.8766. ISSN 0021-9258. PMID 10722720.
- ↑ Kakeya, H.; Onose, R.; Osada, H. (1998-11-01). "Caspase-mediated activation of a 36-kDa myelin basic protein kinase during anticancer drug-induced apoptosis". Cancer Research. 58 (21): 4888–4894. ISSN 0008-5472. PMID 9809995.
- 1 2 Yamada, Yuriko; Taketani, Shigeru; Osada, Hiroyuki; Kataoka, Takao (2011-09-30). "Cytotrienin A, a translation inhibitor that induces ectodomain shedding of TNF receptor 1 via activation of ERK and p38 MAP kinase". European Journal of Pharmacology. 667 (1–3): 113–119. doi:10.1016/j.ejphar.2011.05.072. ISSN 1879-0712. PMID 21663740.