 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-Amino-9-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-3H-purin-6-one | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.012.278 |
| MeSH | Deoxyguanosine |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H13N5O4 | |
| Molar mass | 267.245 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
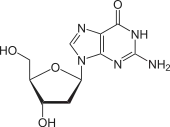
Deoxyguanosine is composed of the purine nucleobase guanine linked by its N9 nitrogen to the C1 carbon of deoxyribose. It is similar to guanosine, but with one hydroxyl group removed from the 2' position of the ribose sugar (making it deoxyribose). If a phosphate group is attached at the 5' position, it becomes deoxyguanosine monophosphate.
Deoxyguanosine is one of the four deoxyribonucleosides that make up DNA.[2]
See also
References
- ↑ "2'-deoxyguanosine". PubChem Compound Database. National Center for Biotechnology Information. 24 December 2016. Archived from the original on 2 January 2017. Retrieved 2 January 2017.
- ↑ "Human Metabolome Database (HMDB) metabocard for Deoxyguanosine (HMDB0000085)".
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.