 | |
| Names | |
|---|---|
| Other names
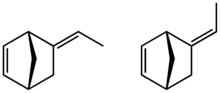
2-ethylidene-5-norbornene | |
| Identifiers | |
PubChem CID |
|
| UNII | |
| Properties | |
| C9H12 | |
| Molar mass | 120.195 g·mol−1 |
| Appearance | colorless liquid |
| Density | 0.893 g/mL |
| Melting point | −80 °C (−112 °F; 193 K) |
| Boiling point | 146 °C (295 °F; 419 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Ethylidene norbornene (ENB) is an organic compound that consists of an ethylidene (CH3C(H)=) group attached to norbornene. It is a colorless liquid. The molecule consists of two sites of unsaturation. The compound consists of E- and Z-stereoisomers, but the mixtures are typically not separated.
Preparation and use
It is prepared by isomerization of vinyl norbornene, which in turn is obtained by the Diels-Alder reaction of butadiene and cyclopentadiene.[1]
It is a monomer that used in the production of the commercial polymer EPDM. Only the ring alkene participates in the copolymerization. The exocyclic double bond (the ethylidene group) undergoes sulfur vulcanization.
Safety
Its LD50 (intravenous, rabbit) ranges from 0.09 (male rabbit) to 0.11 ml/kg (female). It is also a neurotoxin.[2]
References
- ↑ Behr, Arno (2000). "Organometallic Compounds and Homogeneous Catalysis". Ullmann's Encyclopedia of Industrial Chemistry. p. 10. doi:10.1002/14356007.a18_215. ISBN 3527306730.
- ↑ Ballantyne, Bryan; Myers, Roy C.; Klonne, Dennis R. (1997). "Comparative acute toxicity and primary irritancy of the ethylidene and vinyl isomers of norbornene". Journal of Applied Toxicology. 17 (4): 211–221. doi:10.1002/(SICI)1099-1263(199707)17:4<211::AID-JAT430>3.0.CO;2-X. PMID 9285533. S2CID 21154862.