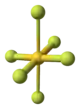
 | |
| Names | |
|---|---|
| IUPAC name
Gold(III) fluoride | |
| Other names
Gold trifluoride Auric fluoride | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| AuF3 | |
| Molar mass | 253.961779 g·mol−1 |
| Appearance | orange-yellow hexagonal crystals |
| Density | 6.75 g/cm3 |
| Melting point | sublimes above 300°C |
| Reacts[2][3] | |
| +74·10−6 cm3/mol | |
| Structure | |
| Hexagonal, hP24 | |
| P6122, No. 178 | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
-363.3 kJ/mol |
| Related compounds | |
Other anions |
Gold(III) chloride Gold(III) bromide |
Other cations |
Silver fluoride Copper(II) fluoride Mercury(II) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Gold(III) fluoride, AuF3, is an orange solid that sublimes at 300 °C.[4] It is a powerful fluorinating agent. It is very sensitive to moisture, yielding gold(III) hydroxide and hydrofluoric acid.
Preparation
Structure
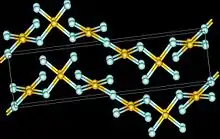
The crystal structure of AuF3 consists of spirals of square-planar AuF4 units.[5]
 |  |  |  |  |
| AuF3 unit cell | neighbouring (AuF3)n helices | distorted octahedral coordination of gold by six fluorines | top-down view of an (AuF3)n helix | side view of an (AuF3)n helix |
References
- ↑ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, Florida: CRC Press. pp. 4–59. ISBN 0-8493-0594-2.
- ↑ Victor Lenher (1903). "Fluoride of Gold.1". Journal of the American Chemical Society. 25 (11): 1136–1138. doi:10.1021/ja02013a004.
- 1 2 Inis C. Tornieporth-Oetting; Thomas M. Klapötke (1995). "Laboratory Scale Direct Synthesis of Pure AuF3". Chemische Berichte. 128 (9): 957–958. doi:10.1002/cber.19951280918.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8., p. 1184.
- ↑ F. W. B. Einstein; P. R. Rao; James Trotter; Neil Bartlett (1967). "The crystal structure of gold trifluoride". Journal of the Chemical Society A: Inorganic, Physical, Theoretical. 4: 478–482. doi:10.1039/J19670000478.
External links
 Media related to Gold trifluoride at Wikimedia Commons
Media related to Gold trifluoride at Wikimedia Commons
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.