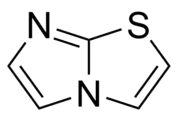
The general structure of imidazothiazoles.
Imidazothiazoles are a class of chemical compounds containing a bicyclic heterocycle (a double ring system) consisting of an imidazole ring fused to a thiazole ring.[1] The structure contains three non-carbon or heteroatoms: two nitrogen atoms and one sulfur atom. Imidazothiazole derivatives show a broad spectrum of in vitro, i.e. "in the petri dish", activity such as anticancer,[2][3][4] antipsychotic,[5] antimicrobial,[6] antifungal,[7] and anthelmintic[8] (against cancer, psychosis, microorganisms, fungi and worms, respectively).
References
- ↑ Fascio, Mirta L.; Errea, María Inés; D'Accorso, Norma Beatriz (2015-01-27). "Imidazothiazole and related heterocyclic systems. Synthesis, chemical and biological properties". European Journal of Medicinal Chemistry. 90: 666–683. doi:10.1016/j.ejmech.2014.12.012. hdl:11336/17776. PMID 25499987.
- ↑ Gürsoy, Elif; Güzeldemirci, Nuray Ulusoy (2007-03-01). "Synthesis and primary cytotoxicity evaluation of new imidazo[2,1-b]thiazole derivatives". European Journal of Medicinal Chemistry. 42 (3): 320–326. doi:10.1016/j.ejmech.2006.10.012. PMID 17145120.
- ↑ Andreani, Aldo; Granaiola, Massimiliano; Leoni, Alberto; Locatelli, Alessandra; Morigi, Rita; Rambaldi, Mirella; Varoli, Lucilla; Lannigan, Deborah; Smith, Jeff; Scudiero, Dominic; Kondapaka, Sudhir (2011-09-01). "Imidazo[2,1-b]thiazole guanylhydrazones as RSK2 inhibitors [1]". European Journal of Medicinal Chemistry. 46 (9): 4311–4323. doi:10.1016/j.ejmech.2011.07.001. hdl:11585/105011. PMID 21794960.
- ↑ Park, Jin-Hun; El-Gamal, Mohammed I.; Lee, Yong Sup; Oh, Chang-Hyun (2011-12-01). "New imidazo[2,1-b]thiazole derivatives: Synthesis, in vitro anticancer evaluation, and in silico studies". European Journal of Medicinal Chemistry. 46 (12): 5769–5777. doi:10.1016/j.ejmech.2011.08.024. PMID 22033063.
- ↑ Cole, Derek C.; Stock, Joseph R.; Lennox, William J.; Bernotas, Ronald C.; Ellingboe, John W.; Boikess, Steve; Coupet, Joseph; Smith, Deborah L.; Leung, Louis; Zhang, Guo-Ming; Feng, Xidong (2007-11-01). "Discovery of N1-(6-Chloroimidazo[2,1-b][1,3]thiazole-5-sulfonyl)tryptamine as a Potent, Selective, and Orally Active 5-HT6 Receptor Agonist". Journal of Medicinal Chemistry. 50 (23): 5535–5538. doi:10.1021/jm070521y. PMID 17948978.
- ↑ Güzeldemirci, Nuray Ulusoy; Küçükbasmacı, Ömer (2010-01-01). "Synthesis and antimicrobial activity evaluation of new 1,2,4-triazoles and 1,3,4-thiadiazoles bearing imidazo[2,1-b]thiazole moiety". European Journal of Medicinal Chemistry. 45 (1): 63–68. doi:10.1016/j.ejmech.2009.09.024. PMID 19939519.
- ↑ Malik, Jitender K.; Soni, Himesh; Singhai, A.K. (2013-01-01). "Synthesis, characterization and evaluation for antifungal activity of substituted diaryl imidazo [2, 1, b]-benzothiazole". Journal of Pharmacy Research. 7 (1): 39–46. doi:10.1016/j.jopr.2013.01.002.
- ↑ Amarouch, Hamid; Loiseau, Philippe R; Bacha, Catarina; Caujolle, Raymond; Payard, Marc; Loiseau, Philippe M; Bories, Christian; Gayral, Philippe (1987-09-01). "Imidazo[2,1-b]thiazoles: analogues du lévamisole". European Journal of Medicinal Chemistry (in French). 22 (5): 463–466. doi:10.1016/0223-5234(87)90037-7.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.