 | |
| Clinical data | |
|---|---|
| Trade names | Metastat (proposed) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
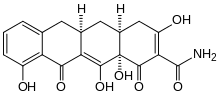
| Formula | C19H17NO7 |
| Molar mass | 371.345 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Incyclinide (proposed trade name Metastat) is a chemically modified tetracycline antibiotic that was investigated in clinical trials for the treatment of rosacea,[1] various tumours, allergic and inflammatory diseases and a number of other conditions.[2][3]
Data from animal studies suggest that centrally infused incyclinide attenuates microglial mediated neuroinflammation in the paraventricular nucleus of the hypothalamus and sympathetic activation in angiotensin II-induced hypertension. This was also associated with unique changes in gut microbial communities and profound attenuation of gut pathology in animal models of hypertension.[4]
Mechanism of action
Like other tetracyclines, incyclinide inhibits matrix metalloproteinases. In contrast to traditional tetracyclines, it lacks antibiotic properties.[5]
References
- ↑ Reuters: CollaGenex says incyclinide ineffective for rosacea
- ↑ Spreitzer H (2 July 2007). "Neue Wirkstoffe - Incyclinid". Österreichische Apothekerzeitung (in German) (14/2007): 655.
- ↑ ClinicalTrials.gov: Incyclinide
- ↑ Sharma RK, Yang T, Oliveira AC, Lobaton GO, Aquino V, Kim S, et al. (March 2019). "Microglial Cells Impact Gut Microbiota and Gut Pathology in Angiotensin II-Induced Hypertension". Circulation Research. 124 (5): 727–736. doi:10.1161/CIRCRESAHA.118.313882. PMC 6395495. PMID 30612527.
- ↑ Viera MH, Perez OA, Berman B (2007). "Incyclinide". Drugs of the Future. 32 (3): 209–214. doi:10.1358/dof.2007.032.03.1083308.