 | |||
| |||
| Identifiers | |||
|---|---|---|---|
3D model (JSmol) |
|||
| ChemSpider | |||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| SiH3I | |||
| Molar mass | 158.014 g/mol | ||
| Appearance | colorless crystals[1] | ||
| Density | 2.070 g·cm−3 (0.5 °C) 2.035 g·cm−3 (14.8 °C)[2] | ||
| Melting point | −56.6 °C (216.6 K)[3] | ||
| Boiling point | 45.8 °C (318.9 K)[3] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
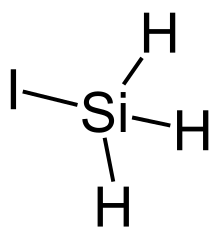
Iodosilane is a chemical compound of silicon, hydrogen, and iodine. It is a colorless monoclinic crystal of space group P21/c at −157 °C.[1]
Preparation
Iodosilane is the first product of the reaction between monosilane and iodine, the other products being di-, tri- and finally tetraiodosilane (silicon tetraiodide).
It can also be produced by the reaction of phenylsilane or chlorophenylsilane with hydrogen iodide.[4]
Properties
At low temperatures, iodosilant quickly reacts with [Co(CO)4]− to form SiH3Co(CO)4.[5]
Further reading
- Nakagawa, Jun; Hayashi, Michiro (1982). "Microwave spectra in the ν3, 2ν3, and ν6 excited states of iodosilane". Journal of Molecular Spectroscopy. 93 (2): 441–444. Bibcode:1982JMoSp..93..441N. doi:10.1016/0022-2852(82)90181-3.
- Ward, Laird G. L.; Norman, Arlan D.; Gondal, S. K.; MacDiarmid, A. G. (1968). "Bromosilane, iodosilane, and trisilylamine". In Jolly, William L.; et al. (eds.). Inorganic Syntheses. Vol. 11. McGraw-Hill. pp. 159–170. doi:10.1002/9780470132425.ch35. ISBN 978-0-470-13170-1.
- Sharbaugh, A. H.; Heath, G. A.; Thomas, L. F.; Sheridan, J. (1953). "Microwave spectrum and structure of iodosilane". Nature. 171 (4341): 87. Bibcode:1953Natur.171...87S. doi:10.1038/171087a0. S2CID 4178870.
References
- 1 2 A. J. Blake, E. A. V. Ebsworth, S. G. D. Henderson, A. J. Welch (1988-08-15). "Structure of silyl iodide at 116 K". Acta Crystallographica Section C Crystal Structure Communications. 44 (8): 1337–1339. doi:10.1107/S0108270188001155. Archived from the original on 2018-06-02. Retrieved 2019-02-25.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ H. J. Emeléus, A. G. Maddock, C. Reid (1941). "68. Derivatives of monosilane. Part II. The iodo-compounds". J. Chem. Soc.: 353–358. doi:10.1039/JR9410000353. ISSN 0368-1769. Retrieved 2019-02-25.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - 1 2 A. G. Maddock, C. Reid, H. J. Emelus (August 1939). "New Iodine and Fluorine Derivatives of Monosilane". Nature. 144 (3642): 328. Bibcode:1939Natur.144Q.328M. doi:10.1038/144328a0. ISSN 0028-0836. S2CID 4118995. Archived from the original on 2021-09-14. Retrieved 2019-02-25.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Handbuch der präparativen anorganischen Chemie / 1 (in German). Stuttgart. p. 686. ISBN 3-432-02328-6. OCLC 310719485.
- ↑ B. J. Aylett, J. M. Campbell (1965). "A volatile silicon–transition-metal compound". Chem. Commun. (London) (11): 217. doi:10.1039/C19650000217. ISSN 0009-241X.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.