| Gnathostomiasis | |
|---|---|
| Other names | Gnathostoma, Larva migrans profundus, Nodular migratory eosinophilic panniculitis, Spiruroid larva migrans, Wandering swelling, Yangtze edema[1] |
| Specialty | Infectious diseases, helminthology |
Gnathostomiasis, also known as larva migrans profundus,[2]: 436 is the human infection caused by the nematode Gnathostoma spinigerum and/or Gnathostoma hispidum, which infects vertebrates.
Symptoms and signs
A few days after ingestion epigastric pain, fever, vomiting, and loss of appetite resulting from migration of larvae through intestinal wall to the abdominal cavity will appear in the patient.[3] Migration of parasites in the subcutaneous tissues causing intermittent, migratory, painful, pruritic swellings is known as cutaneous larva migrans. Patches of edema appear after initial symptoms clear and are usually found on the abdomen.[3] These lesions vary in size and can be accompanied by pruritus, rash, and stabbing pain. Swellings may last for 1 to 4 weeks in a given area and then reappear in a different location.[3] Migration to other tissues causes visceral larva migrans and can result in cough, hematuria, ocular involvement,[4] meningitis, encephalitis and eosinophilia. Eosinophilic myeloencephalitis may also result from invasion of the central nervous system by the larvae.[1]
Causes
Human gnathostomiasis is infection by the migrating third-stage larvae of any of five species of Gnathostoma. The most common cause in Asia is G. spinigerum, and the most common cause in the Americas is G. binucleatum. G. hispidium and G. doloresi occur in East and Southeast Asia; the former has also been found in Eastern Europe. G. nipponicum occurs only in Japan and China.[5][6][7] There is one unconfirmed report of G. malaysiae causing disease in humans.[8]
Transmission
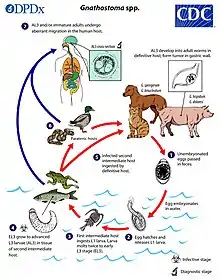
Gnathostomiasis is transmitted by the ingestion of raw or insufficiently cooked definitive hosts such as fresh water fish, poultry, or frogs.
In Thailand and Vietnam, the most common cause appears to be consumption of undercooked Asian swamp eels (Monopterus albus, also called Fluta alba) which transmit G. spinigerum.[9][10][11][12] Monopterus albus is an invasive species in North America, but no Gnathostoma infections in humans have yet been conclusively identified in the US.[13]
Hosts
Intermediate host
The primary intermediate host is the minute crustaceans of the genus Cyclops.[14] These crustaceans are then ingested by a second intermediate host, such as frogs.
Definitive host
The definitive hosts for gnathostomiasis include cats, dogs, tigers, leopards, lions, mink, opossums, raccoons, poultry, frogs, freshwater fish, snakes or birds.[1]
Incubation period
The incubation period for gnathostomiasis is 3–4 weeks when the larvae begin to migrate through the subcutaneous tissue of the body.[15]
Morphology
The adult parasite is reddish-brown in color and has a globular cephalic dome that is separated from the rest of the body by constriction.[15] The posterior portion of the nematode is smooth while the anterior half is covered with fine leaf-like spines.[15] The head is round and contains 4 to 8 transverse rows of hooklets that are protected by a pair of fleshy lips . The males are shorter than the females, 11–25 mm (0.43–0.98 in) compared to 25–54 mm (0.98–2.13 in) respectively.[15] Eggs are oval and have a mucoid plug at one end.[15]
Life cycle
Life cycle in definitive hosts
Adult worms are found in a tumor located in the gastric wall of the definitive hosts and release eggs into the host's digestive tract. The eggs are then released with feces and in about a week hatch in water to develop into first stage larva.[16] Larvae are then ingested by minute copepods of the genus Cyclops.[17] Once entering the copepod, the larvae penetrate the gastric wall of their intermediate host and begin to develop into second-stage and even early third-stage larvae.[16] The copepods are then ingested by a second intermediate host such as fish, frogs, or snakes.[3] Within this second intermediate or definitive host the larva repeat a similar pattern of penetrating the gastric wall, but then continue to migrate to muscular tissue and develop into advanced third-stage larvae.[16] These larvae then encyst within the musculature of the new host.[18] If the cyst containing flesh of these hosts is ingested by a definitive host, such as dogs, and cats, the cysts are ingested and the larvae escape the cysts and penetrate the gastric wall.[16] These released larvae travel to the connective tissue and muscle as observed before and after 4 weeks they return to the gastric wall as adults.[16] Here they form a tumor and continue to mature into adults for the next 6–8 months.[3] Worms mate and females begin to excrete fertilized eggs with feces 8–12 months after ingestion of cysts.[14] They are passed out in the feces and eaten by another fish.
Life cycle in humans
Infection of humans by gnathostomiasis is accidental because humans are not one of the definitive hosts of the parasite and do not allow the parasite to complete its life cycle. Infection in humans follows ingestion of raw or insufficiently cooked infected intermediate hosts.[14] The ingested third stage larva migrates from the gastric wall and its migration results in the symptoms associated with infection by gnathostomiasis.[3] The third stage larvae don't return to the gastric wall preventing it from maturing into adult worms, leaving the life cycle incomplete. Instead the larvae continue to migrate unpredictably unable to develop into adults, so eggs are seldom found in diagnostic tests.[1] This also means the number of worms present in humans is a reflection of the number of third stage larvae ingested.
Diagnosis
Diagnosis of gnathostomiasis is possible (with microscopy) after removal of the worm. The primary form of diagnosis of gnathostomiasis is the identification of larva in the tissue.[1] Serological testing such as enzyme-linked immunosorbent assay (ELISA) or the Western blot are also reliable but may not be easily accessible in endemic areas.[1]
CT scanning or MRI can be used to help identify a soft tissue worm and when looking at CNS disease it can be used to reveal the presence of the worm.[3] The presence of haemorrhagic tracks on gradient-echo T2-weighted MRI is characteristic and possibly diagnostic.[6]
Prevention
The best strategies for preventing accidental infection of humans is to educate those living in endemic areas to only consume fully cooked meat. The inability of the parasite to complete its life cycle within humans means that transmission can easily be contained by adequate preparation of meat from intermediate hosts. This is especially useful because of the difficulty and lack of feasibility inherent in eliminating all intermediate hosts of gnathostomiasis. So instead, individuals in endemic areas should avoid eating raw and undercooked meat in endemic areas, but this may be difficult in these areas.
The dish ceviche is native to Peru and a favorite of Mexico. It consists of onion, cubed fish, lime or lemon juice and Andean spices including salt and chili. The ingredients are mixed together and they are allowed to marinate several hours before being served at room temperature. Then in endemic areas in Southeast Asia there are traditional dishes associated with these areas that also include raw uncooked fish, such as koipla in Thailand, goi ca song in Vietnam, sashimi and sushi in Japan.[16]
Acknowledging these cultural traditions, individuals in these cultural can be educated on methods of adapting their food preparation activities in order to remove the larvae without greatly altering these traditional dishes. For instance, meat should be marinated in vinegar for six hours or in soy sauce for 12 hours in order to successfully kill the larvae. In areas with reliable electricity, meat can be frozen at -20 degrees Celsius for 3–5 days to achieve the same results of killing the larvae present.[14]
Treatment
Surgical removal or treatment with albendazole or ivermectin is recommended. The most prescribed treatment for gnathostomiasis is surgical removal of the larvae but this is only effective when the worms are located in an accessible location.[1] In addition to surgical excision, albendazole and ivermectin have been noted in their ability to eliminate the parasite.[3] Albendazole is recommended to be administered at 400 mg daily for 21 days as an adjunct to surgical excision, while ivermectin is better tolerated as a single dose.[1] Ivermectin can also serve as a replacement for those that can't handle albendazole 200 ug/kg p.o. as a single dose.[1] However, ivermectin has been shown to be less effective than albendazole.[19]
Epidemiology
Endemic areas include Asia, Mexico, India and parts of South Africa.[3] Originally believed to be confined to Asia, in the 1970s gnathostomiasis was discovered in Mexico,[3] and found in Australia in 2011.[20][21] Even though it is endemic in areas of Southeast Asia and Latin America, it is an uncommon disease. However, researchers have noticed recently an increase in incidence. This disease is most common in both Thailand and Japan, but in Thailand it is responsible for most of the observed parasitic CNS infection.[14] It has long been recognised in China, but reports have only recently appeared in the English literature.[22]
History
The first case of Gnathostoma infection was identified by Sir Richard Owen when inspecting the stomach of a young tiger that had died at London Zoo from a ruptured aorta.[23] However it was not until 1889 that the first human case was described by Levinson when he found the Gnathostoma larva in an infested Thai woman. The lifecycle of G. spinigerum was described by Svasti Daengsvang and Chalerm Prommas from Thailand in 1933 and 1936.[24] This delay in identification of the parasite in humans is due to the fact that humans are not a definitive host for this parasite making infection from this parasite rare. Gnathostomiasis infection is rare because the parasite must be digested when it has reached its third larvae stage, providing only a short time frame in which the parasite is capable of infecting humans. It is uncommon for the larvae to penetrate the skin of individuals exposed to contaminated food or water without ingestion.[14]
See also
References
- 1 2 3 4 5 6 7 8 9 Gideon; Gnathostomiasis
- ↑ William D. James; Timothy G. Berger; Dirk M. Elston (2006). Andrews' Diseases of the Skin: Clinical Dermatology (10th ed.). Saunders Elsevier. ISBN 0-7216-2921-0.
- 1 2 3 4 5 6 7 8 9 10 David T. John; William A. Petri (2006). "The blood- and tissue-dwelling nematodes". Markell and Voge's Medical Parasitology (9th ed.). Elsevier. pp. 274–321. ISBN 978-0-7216-4793-7.
- ↑ P. Barua; N. K. Hazarika; N. Barua; C. K. Barua; B. Choudhury (2007). "Gnathostomiasis of the anterior chamber". Indian Journal of Medical Microbiology. 25 (3): 276–8. doi:10.1016/S0255-0857(21)02122-8. hdl:1807/53473. PMID 17901651.
- ↑ "CDC - DPDx - Gnathostomiasis". www.cdc.gov. 2019-05-07. Retrieved 2019-08-14.
- 1 2 Katchanov J, Sawanyawisuth K, Chotmongkol V, Nawa Y (2011). "Neurognathostomiasis, a neglected parasitosis of the central nervous system". Emerging Infectious Diseases. 17 (7): 1174–1180. doi:10.3201/eid1707.101433. PMC 3321562. PMID 21762569.
- ↑ Herman JS, Chiodini PL (2009). "Gnathostomiasis, another emerging imported disease". Clin Microbiol Rev. 22 (3): 484–92. doi:10.1128/CMR.00003-09. PMC 2708391. PMID 19597010.
- ↑ Nomura Y, Nagakura K, Kagei N, Tsutsumi Y, Araki K, Sugawara M (2000). "Gnathostomiasis possibly caused by Gnathostoma malaysiae". Tokai J Exp Clin Med. 25 (1): 1–6. PMID 11023048.
- ↑ Setasuban P; Nuamtanong S; Rojanakittikoon V; Yaemput S; Dekumyoy P; Akahane H; Kojima S (1991). "Gnathostomiasis in Thailand: a survey on intermediate hosts of Gnathostoma spp. with special reference to a new type of larvae found in Fluta alba". Southeast Asian J Trop Med Public Health. 22 Suppl: 220–4. PMID 1822890.
- ↑ Akahane H, Setasuban P, Nuamtanong S, Horiuchi S, Koga M, Kojima S (1995). "A new type of advanced third-stage larvae of the genus Gnathostoma in freshwater eels, Fluta alba, from Nakhon Nayok, central Thailand". Southeast Asian J Trop Med Public Health. 26 (4): 743–7. PMID 9139388.
- ↑ Saksirisampant W, Kulkaew K, Nuchprayoon S, Yentakham S, Wiwanitkit V (Mar 2002). "A survey of the infective larvae of Gnathostoma spinigerum in swamp eels bought in a local market in Bangkok, Thailand". Ann Trop Med Parasitol. 96 (2): 191–5. doi:10.1179/000349802125000295. PMID 12080980. S2CID 24447639.
- ↑ Sieu TP, Dung TT, Nga NT, Hien TV, Dalsgaard A, Waikagul J, Murrell KD (Feb 2009). "Prevalence of Gnathostoma spinigerum infection in wild and cultured swamp eels in Vietnam". J Parasitol. 95 (1): 246–8. doi:10.1645/GE-1586.1. PMID 19245276. S2CID 23748298.
- ↑ Nico LG, Paul Sharp P, Collins TM (2011). "Imported Asian swamp eels (Synbranchidae: Monopterus) in North American live food markets: potential vectors of non-native parasites". Aquatic Invasions. 6 (1): 69–76. doi:10.3391/ai.2011.6.1.08.
- 1 2 3 4 5 6 Robert W. Tolan Jr. (January 2009). "Gnathostomiasis". Medscape. Retrieved July 8, 2011.
- 1 2 3 4 5 G. N. Seal; A. K. Gupta; M. K. Das (1969). "Intra-ocular gnathostomiasis". Indian Journal of Ophthalmology. 17 (3): 109–13. PMID 5392612.
- 1 2 3 4 5 6 K. Darwin Murrell; Bernard Fried (2007). Food-Borne Parasitic Zoonoses, Fish and Plant-Borne Parasites. World Class Parasites. Vol. 11. Springer. doi:10.1007/978-0-387-71358-8. ISBN 978-1-4419-4392-7.
- ↑ "CDC Life Cycle of Gnathostomiasis". Archived from the original on 2013-02-15. Retrieved 2009-02-26.
- ↑ Rebecca A. Cockman-Thomas; Curtis A. Colleton; Chris H. Gardiner; Wayne M. Meyers (1993). "Gnathostomiasis in a wild-caught nine-banded armadillo (Dasypus novemcinctus)". Laboratory Animal Science. 43 (6): 630–2. PMID 8158995.
- ↑ Thiangtrongjit, Tipparat; Nogrado, Kathyleen; Ketboonlue, Thawatchai; Malaitong, Preeyarat; Adisakwattana, Poom; Reamtong, Onrapak (2021-08-25). "Proteomics of Gnathostomiasis: A Way Forward for Diagnosis and Treatment Development". Pathogens. 10 (9): 1080. doi:10.3390/pathogens10091080. ISSN 2076-0817. PMC 8465481. PMID 34578113.
- ↑ Cameron J Jeremiah; Chanad S Harangozo; Andrew J Fuller (2011). "Gnathostomiasis in remote northern Western Australia: the first confirmed cases acquired in Australia". Medical Journal of Australia. 195 (1): 42–44. doi:10.5694/j.1326-5377.2011.tb03188.x. PMID 21728942. S2CID 37139535.
- ↑ "Couple eaten alive by tiny worms". Yahoo. July 5, 2011. Retrieved July 8, 2011.
- ↑ Dong Ming Li; Xue Rong Chen; Jing Song Zhou; Zhi Biao Xu; Yukifumi Nawa; Paron Dekumyoy (2009). "Short report: case of gnathostomiasis in Beijing, China" (PDF). American Journal of Tropical Medicine and Hygiene. 80 (2): 185–7. doi:10.4269/ajtmh.2009.80.185. PMID 19190210.
- ↑ Yangtse Oedema (Gnathostomiasis) Patient UK
- ↑ Valai Bussaratid; Srivicha Krudsood; Udomsak Silachamroon; Sornchai Looareesuwan (2005). "Tolerability of ivermectin in gnathostomiasis" (PDF). Southeast Asian Journal of Tropical Medicine and Public Health. 36 (3): 644–9. PMID 16124431.
External links
- Gnathostomiasis Archived 2013-02-15 at the Wayback Machine at CDC
- Gnathostomiasis at eMedicine