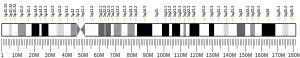Methionine synthase reductase, also known as MSR, is an enzyme that in humans is encoded by the MTRR gene.[5][6]
Function
Methionine is an essential amino acid required for protein synthesis and one-carbon metabolism. Its synthesis is catalyzed by the enzyme methionine synthase. Methionine synthase eventually becomes inactive due to the oxidation of its cobalamin cofactor. Methionine synthase reductase regenerates a functional methionine synthase via reductive methylation. It is a member of the ferredoxin-NADP(+) reductase (FNR) family of electron transferases.[6]
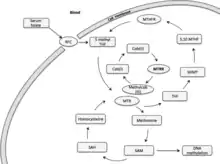
Methionine synthase reductase (MTRR) is primarily involved in the reductive methylation of homocysteine to methionine, utilizing methylcob(I)alamin as an intermediate methyl carrier.[7] Methionine is an essential amino acid in mammals, necessary for protein synthesis and one carbon metabolism. In its activated form, S-adenosylmethionine acts as a methyl donor in biological transmethylation reactions and as a propylamine donor in polyamine synthesis.[7] A major product of methionine demethylation is homocysteine. Remethylation of homocysteine occurs via a cobalamin dependent enzyme, methionine synthase (MTR).[7] The folate cycle is linked to homocysteine metabolism via MTR.[8] Circulating blood folate (5-methyl tetrahydrofolate, 5-MTHF) donates methyl groups to MTR to be utilized in cellular methylation. A methyl cobalt bond of the intermediary methyl carrier, methlycob(III)alamin is cleaved heterolytically producing cobalamin in its highly reactive oxidation state as cob(I)alamin. The enzyme bound cob(I)alamin cofactor of the MTR enzyme functions as a methyl carrier between 5-MTHF and homocysteine.[7] Cob(I)alamin is oxidised to cob(II)alamin about once every 100 methyl transfer cycles, rendering the cob(I)alamin-MTR-enzyme complex inactive.[9] Reactivation of this enzyme complex occurs through reductive remethylation by MTRR, utilizing S-adenosylmethionine as a methyl donor. MTR reactivation can also be NADPH dependent involving two redox proteins, soluble cytochrome b5 and reductase 1. However, this pathway is responsible for a minor role in reactivation, whilst MTRR remains a major contributor in this reductive reactivation.
Biological processes influenced by MTRR include: sulfur amino acid metabolic process, DNA methylation, methionine metabolic process, methionine biosynthetic process, methylation, S-adenosylmethionine cycle, homocysteine catabolic process, folic acid metabolic process, oxidation-reduction process and negative regulation of cystathionine beta-synthase activity.[7][8]
MTRR gene
The Methionine Synthase Reductase (MTRR) gene primarily acts in the reductive regeneration of cob(I)alamin (vitamin B12).[10] Cob(I)alamin is a cofactor that maintains activation of the methionine synthase enzyme (MTR) Methionine synthase, linking folate and methionine metabolism. Donation of methyl groups from folate are utilized for cellular and DNA methylation, influencing epigenetic inheritance.[10][7][11]
Aliases
- 5-Methyltetrahydrofolate-Homocysteine Methyltransferase Reductase
- MSR
- [Methionine Synthase]- Cobalamin Methyltransferase (Cob(II)Alamin Reducing)
- Methionine Synthase Reductase, Mitochondrial
- EC 1.16.1.8
- CblE
Mapping
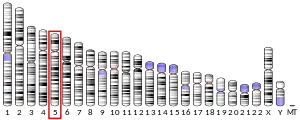
The gene was mapped to human chromosome 5. Gene specific primer pairs resulted in PCR amplification of a product matched by size to a hybrid-mapping panel containing only chromosome 5 as its human genetic material.[5] The product DNA sequence matched pre-established gene markers specific to this chromosome. Exact gene cytogenic position was determined by mapping to an artificial chromosomal construct containing the gene via fluorescence in situ hybridization.[5] The exact MTRR gene location was mapped to 5p15.3-p15.2.[5]
Structure
The MTRR gene is associated with a family of electron transferases known as the Ferredoxin-NADP(+) reductase (FNR) family. Found in 15 primates and over 16 tissues in humans, MTRR is 34 kb long.[12] The gene comprises 15 exons and includes numerous cytolosic mitochondrial mRNA isoforms. Multiple cofactor binding sites assist in the maintenance of MTR activity via reductive remethylation. All binding domains involve selective and non-covalent interactions except the flavodoxin_1 domain.[12]
Co-factor binding sites
Flavodoxin-1 domain
Flavoproteins are ubiquitous biocatalysts binding specific redox active prosthetic groups. The domain is associated with electron transfer proteins and used in electron transport systems.[12] The cofactor flavin-mononucleotide (FMN) is bound non-covalently to the domain, which is functionally interchangeable with iron-sulfur constituted proteins regulating electron transfer or ferredoxins.[12]
FAD
Flavin adenine dinucleotide in its oxidized form, FAD is a cofactor of flavoprotein oxidoreductase enzymes. Flavoprotein pyridine nucleotide cytochrome reductases, including FAD catalyse the interchange of reducing equivalents (H+ or electrons). Initial electron donors and final electron acceptors comprise single electron carriers and two electron carrying nicotinamide dinucleotides respectively.[12]
NAD
Evolutionary conserved protein domain corresponding to oxidoreductase activity. NAD binding catalyzes redox reactions to alter the oxidation state of metal ions, using NADP+ as an electron acceptor.[12]
Methionine synthase reductase activity
Involved in the reductive remethylation of cob(II)alamin using S-adenosylhomocysteine as a methyl donor. Catalyses the reaction:
[methionine synthase]- cob(II)alamin + NADPH + H+ + S-adenosylmethionine → [methionine synthase]-methylcob(I)alamin + S-adenosylhomocysteine + NADP+.[12]
Aquacobalamin reductase (NADPH) activity
Belongs to the oxidoreductase family, oxidizing metal ions with NADP+ acting as an electron acceptor. Uses FAD as a cofactor when catalyzing the following reaction:
2cob(II)alamin + NADP+ 2aquacob(III)alamin + NADPH + H+.[12]
Flavin adenine dinucleotide
Interacts with the cofactor or prosthetic group, FAD of flavoproteins and contains a flavin moiety in the form of FAD or FMN (flavin mononucleotide). The domain non-covalently binds oxidized FAD or its reduced form, hydroquinone (FADH2).[12]
FMN
Flavin mononucleotide binding domain interacts with a coenzyme of flavoprotein oxidoreductase enzymes, FMN.[12]
NADP
Nicotinamide adenine dinucleotide phosphate is a coenzyme present in redox and biosynthetic reactions. The domain binds NADP in its oxidised or reduced forms as NADP+ or NADPH respectively.[12]
ADPH
Involves a non-covalent and selective interaction with the reduced form of NADP, NADPH.[12]
NADPH-hemoprotein reductase activity
The domain is associated with the oxidoreductase family and acts on NADH or NADPH, using a heme protein as an electron acceptor. Requires FAD and FMN as cofactors to catalyse the reaction:
NADPH + H+ + n oxidised hemoprotein = NADP+ + n reduced hemoprotein.[12]
Protein binding
Binding domain involved in the interaction with proteins or protein complexes.[12]
Polymorphisms
Pathogenic mutations
- (MTRR):c.66A>G – Polymorphism resulting in isoleucine conversion to methionine at codon 22. This mutation is found within and affects the FMN binding domain.[13]
- (MTRR):c.524C>T – Serine to leucine substitution at codon 175. Benign mutation associated with impaired intracellular cobalamin metabolism disorders.[14]
- (MTRR):c.1049A>G – Lysine to arginine substitution at codon 350.[15]
- (MTRR):c.1349C>G – Proline to arginine substitution at codon 450. Prevalence associated with abnormal intracellular cobalamin metabolism disorders.[16]
- (MTRR):c.903+469T>C – Deep intronic insertion between exons 6 and 7 (r.903_904ins140). Threonine to cysteine change resulting in activation of an exon splicing enhancer in intron 6.[17]
- (MTRR):c.1361C>T – Rare polymorphism involving serine to leucine substitution at codon 454. Known as the Iberian mutation, prevalent in homocystinuria megaloblastic anemia due to impaired cobalamin metabolism. Mainly three different halotypes (GTACG, GCACA, GCACG) from the deamination of methyl cytosine in different chromosomes.[18]
- (MTRR):c.1459G>A – Involves glycine to arginine substitution at codon 487. Conserved in MTRR and found to occur within the FAD binding domain. Pathogenicity associated with inborn genetic diseases.[19]
- (MTRR):c.1573C>T – Arginine substitution with a premature termination codon at codon 525.[20]
- (MTRR):c.1622_1623dupTA – Results in formation of a premature termination codon. Pathogenicity associated with CblE type of homocystinuria.[21]
- (MTRR):r.1462_1557del96 – Associated with splicing of exon 11 due to a 7 base pair deletion. A large deletion of this mutant allele results in the absence of a c-terminus in the FAD binding domain. Pathogenicity associated with CblE type of homocystinuria.[21]
- (MTRR):c.1953-6_1953-2del5 – Novel mutation associated with CblE type of homocystinuria. Unstable mRNA arising from this mutant results in an absence of mRNA required for translation, producing pathogenicity.[21]
Mutations involved in the formation of premature termination codons result in truncated mutated proteins if translated.[21] Mutants exhibit an absence of FAD/NADPH binding domains and unstable mRNA due to nonsense mediated decay (NMD). NMD is not present in (MTRR):c.1573C>T or (MTRR):c.1622_1623dupTA polymorphisms.[21] The (MTRR):c.903+469T>C variant is also associated with the formation of premature termination codons.[21]
Other mutations
- (MTRR):c.1911G>A ¬– Benign synonymous mutation (alanine to alanine) at codon 637. Associated with disorders of cobalamin metabolism.[22]
Clinical significance
Single nucleotide polymorphisms (SNPs) in the MTRR gene impair MTR activity, resulting in elevated homocysteine levels due to compromised methylation to methionine. Elevated homocysteine levels are associated with birth defects in addition to pregnancy complications, cardiovascular disease, cancer,[23] megaloblastic anemia, Alzheimer’s diseases and cognitive dysfunction in the elderly.[24] Presence of the mutant variant (66A>G) is associated with significantly lower, up to 4 fold, plasma cobalamin and folate levels in cardiac transplant patients.[23] A consequent decrease in S-adenosylmethionine availability results in DNA hypomethylation. Low folate limits one carbon metabolism and homocysteine metabolism as vitamin B12 interacts with folate in this pathway.[24] Additionally, this mutation is associated with an increased risk in type 2 diabetes.[25]
Cancer
Specific SNPs are associated with an elevated risk of lung cancer and interact with folate dietary intake in this etiology. The (MTRR):c.66A>G mutation relates to a significant increase in the risk of lung cancer. When co-expressed with the polymorphism (MTR): 2756A>G, lung cancer risk is further increased in a dose dependent manner.[24] Correlation between this polymorphism and increased risk of lung cancer is present with low folate intake and high vitamin B12, suggesting a B12 independent mechanism of action.[24] This mutation is also associated with an increased risk in colorectal cancer,[26][27][28][29] acute lymphoblastic leukemia,[30] bladder cancer,[31] cervical intraepithelial neoplasia, non-Hodgkin lymphoma[32] and oesophageal squamous cell carcinoma.[33]
CblE type of homocystinuria
Remethylation of homocysteine to methionine by MTR requires the derivative of cobalamin, methylcobalamin. Cobalamin metabolism is initiated by the endocytosis of cobalamin bound to the plasma protein transcobalamin (II). Cleavage of this complex produces free cobalamin, translocating from lysosome to cytoplasm. Conversion can occur to 5’-deoxyadenosylcobalamin (AdoCbl) activating the mitochondrial enzyme methylmalonly coenzyme A mutase or to methylcobalamin (MeCbl).[34] An error in cobalamin metabolism resulting in decreased MeCbl and unaffected AdoCbl is characteristic of the CblE type of homocystinuria.[34] This complementation is rare with autosomal recessive inheritance. The inherited methionine synthase functional deficiency corresponds to a defect in the reducing system required to activate the MTR enzyme. Symptoms of this condition comprise developmental retardation, megaloblastic anemia, homocystinuria, hypomethioninemia, cerebral atrophy and hyperhomocysteinemia.[34] However, hypomethioninemia remains an inconsistent symptom. Decreased MeCbl alongside normal cobalamin uptake is suggestive of decreased intracellular methionine biosynthesis. Occurring mainly in childhood, 15 pathogenic mutations can be associated with CblE type homocystinuria.[35] Additionally, vascular abnormalities are associated with this defect.[36] Impaired reduction of an oxidised cobalt atom in the active site of MTR is associated with this condition, where enzyme activity can be corrected with reducing agents.[35] Rare polymorphisms related to this disease include (MTRR):c.1459G>A, (MTRR):c.1623-1624insTA and (MTRR):c.903+469T>C.[35] These mutations, excluding (MTRR):c.1459G>A result in a frame shift, producing premature termination codons.[35] As consequent products are distant from normal, mutant mRNA arises and nonsense mediated decay (FMN) is initiated.[35] The large insertion of 903_904ins140 corresponding to 903+469T>C is most prevalent in CblE pathology. The activation of an enhanced splicer within intron 6 is incomplete, producing small quantities of normal spliced MTRR mRNA.[35] Prenatal diagnosis of this condition is possible using [14C] methyltetrahydrofolate.[35] Mutation analysis in native chorionic villi and [14C] formate in amino acids within these villi or cultured amniocytes is indicative of the CblE defect.[35] Additionally, macrocytic anemia is a typical feature of the CblE defect and can be corrected though OH-Cobalamin administration or folate supplementation.[35]
Coronary artery disease
Homocysteine, a sulfur based amino acid is the main product of methionine demethylation. Elevated homocysteine is an independent risk factor for cardiovascular disease and inversely correlated to consumed vitamin B12/B6 and folate levels.[37] Homocysteine methylation to methionine is catalyzed by MTR, resulting in appropriate intracellular levels of methionine and tetrahydrofolate, alongside non-toxic homocysteine levels. The GG phenotype promotes the development of premature coronary artery disease (CAD) independent of hyperhomocysteinemia.[37] Hyperhomocysteinemia is associated with cerebral, coronary and peripheral atherosclerotic pathology as it promotes endothelial cell dysfunction, platelet adhesion and vascular smooth muscle cell proliferation.[36] DNA damage and homocysteine levels are proportional to CAD severity. Micronucleus frequency in human lymphocytes, dependent on homocysteine levels increases reactive oxygen species and uracil incorporation in DNA methylation, promoting genetic alterations and point mutations.[36] The (MTRR):c.66A>G, polymorphism containing chromosome is prone to fragmentation. This chromosomal loss or global DNA hypomethylation results in under condensation of pericentromeric heterochromatin, micronucleus formation and elevated risks of aneuploidy.[36] Co-expression of this mutation and the 677T polymorphism in methionine tetrahydrofolate reductase (MTHFR) Methylenetetrahydrofolate reductase act to further the extent of DNA damage.[36]
Hypomethylation due to impaired methylation up regulates atherosclerotic susceptible genes whilst down regulating atherosclerosis protective genes.[36] This abnormality is present during the atherosclerotic pathology, increasing transcriptional activity of platelet derived growth factor (PDGF) and promoting smooth muscle cell proliferation.[36]
Neural tube defects
Spina bifida
MTRR requires vitamin B12 for maintenance of the methyl synthase reaction whilst folate is needed for normal synthesis of nucleotide precursors. These ensure normal DNA synthesis and cellular methylation reactions.[7] Chronic folate or methyl deficiencies are thereby linked to abnormal DNA methylation. The 66A>G polymorphism is up-regulated in neural tube defects and increases the risk of spina bifida by two-fold.[38] Homozygosis for this mutation is an established maternal risk factor for spina bifida especially with low intracellular vitamin B12[39] in the circulation or amniotic fluid.[13] Vitamin B12 is reflected by plasma methylmalonic acid (MMA), an elevation in which indicates impaired B12 uptake or metabolism.[7] A raised MMA combined with the MTRR mutation corresponds to a 5-fold increase in spina bifida.[7] The mechanism of action of this polymorphism is through the mother, hence there is no preferential transmission of this mutation from parent to child. Abnormal MTRR binding to the MTR-cob(I)alamin-enzyme complex down regulates the rate of homocysteine methylation. Consequent decreases in methionine and S-adenosylmethionine negatively affect DNA, gene and protein methylation, all of which are involved in neural tube closure.[7] Increased proliferation during neurulation decreases the availability of DNA nucleotides. As these are unable to be replaced due to impaired DNA methylation and nucleotide formation, consequent disturbed neurulation results in the formation of neural tube defects.[11] Co-expression of this mutation with the 677C>T MTHFR polymorphism furthers the risk of spina bifida compared to an independent acting 66A>G mutation.[7]
Down syndrome
Trisomy 21 or Down syndrome is the most common human chromosomal anomaly arising from abnormal chromosomal segregation in meiosis.[10][11][40] The condition can occur during anaphase in meiosis(I) marking oocyte maturation before ovulation and/or during anaphase in meiosis (II) signifying fertilization.[11] Metabolic impact during these stages is furthered by low vitamin B12.[41] Methylation of homocysteine to methionine is affected, primarily by the (MTRR):c.66A>G polymorphism. Chronic homocysteine elevation increases s-adenosyl-L-homocysteine levels, consequently inhibiting methyltransferase activity and promoting DNA hypomethylation.[10][42] Mothers homozygous for this mutation (GG phenotype) are at a greater risk of having a child with down syndrome compared to heterozygotes (GA phenotype).[41] Geographically, Irish populations are more likely to be homogenous whilst north American populations are commonly heterogeneous, resulting in a greater incidence of the polymorphism in the former group.[11][41] The homozygous mutant allele promotes DNA hypomethylation and meiotic non-disjunction, increasing the risk of down syndrome.[43] This polymorphism correlates to a 2.5 fold risk increase independently and a 4 fold increase in risk when co-expressed with the 677C>T MTHFR mutation.[11] Combination with the MTR2756A>G genetic polymorphism further elevates down syndrome risk.[11][42][44]
See also
References
- 1 2 3 GRCh38: Ensembl release 89: ENSG00000124275 - Ensembl, May 2017
- 1 2 3 GRCm38: Ensembl release 89: ENSMUSG00000034617 - Ensembl, May 2017
- ↑ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- 1 2 3 4 Leclerc D, Wilson A, Dumas R, Gafuik C, Song D, Watkins D, Heng HH, Rommens JM, Scherer SW, Rosenblatt DS, Gravel RA (March 1998). "Cloning and mapping of a cDNA for methionine synthase reductase, a flavoprotein defective in patients with homocystinuria". Proceedings of the National Academy of Sciences of the United States of America. 95 (6): 3059–64. Bibcode:1998PNAS...95.3059L. doi:10.1073/pnas.95.6.3059. PMC 19694. PMID 9501215.
- 1 2 "Entrez Gene: MTRR 5-methyltetrahydrofolate-homocysteine methyltransferase reductase".
- 1 2 3 4 5 6 7 8 9 10 11 van der Linden IJ, den Heijer M, Afman LA, Gellekink H, Vermeulen SH, Kluijtmans LA, Blom HJ (December 2006). "The methionine synthase reductase 66A>G polymorphism is a maternal risk factor for spina bifida". Journal of Molecular Medicine. 84 (12): 1047–54. doi:10.1007/s00109-006-0093-x. PMID 17024475. S2CID 24500664.
- 1 2 Zhang T, Lou J, Zhong R, Wu J, Zou L, Sun Y, Lu X, Liu L, Miao X, Xiong G (2013). "Genetic variants in the folate pathway and the risk of neural tube defects: a meta-analysis of the published literature". PLOS ONE. 8 (4): e59570. Bibcode:2013PLoSO...859570Z. doi:10.1371/journal.pone.0059570. PMC 3617174. PMID 23593147.
- ↑ Vaughn JD, Bailey LB, Shelnutt KP, Dunwoody KM, Maneval DR, Davis SR, Quinlivan EP, Gregory JF, Theriaque DW, Kauwell GP (November 2004). "Methionine synthase reductase 66A->G polymorphism is associated with increased plasma homocysteine concentration when combined with the homozygous methylenetetrahydrofolate reductase 677C->T variant". The Journal of Nutrition. 134 (11): 2985–90. doi:10.1093/jn/134.11.2985. PMID 15514263.
- 1 2 3 4 Wang SS, Qiao FY, Feng L, Lv JJ (February 2008). "Polymorphisms in genes involved in folate metabolism as maternal risk factors for Down syndrome in China". Journal of Zhejiang University Science B. 9 (2): 93–9. doi:10.1631/jzus.B0710599. PMC 2225490. PMID 18257130.
- 1 2 3 4 5 6 7 Hobbs CA, Sherman SL, Yi P, Hopkins SE, Torfs CP, Hine RJ, Pogribna M, Rozen R, James SJ (September 2000). "Polymorphisms in genes involved in folate metabolism as maternal risk factors for Down syndrome". American Journal of Human Genetics. 67 (3): 623–30. doi:10.1086/303055. PMC 1287522. PMID 10930360.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 "MTRR - Methionine synthase reductase - Homo sapiens (Human) - MTRR gene & protein". Retrieved 2017-09-16.
- 1 2 Wilson A, Platt R, Wu Q, Leclerc D, Christensen B, Yang H, Gravel RA, Rozen R (August 1999). "A common variant in methionine synthase reductase combined with low cobalamin (vitamin B12) increases risk for spina bifida". Molecular Genetics and Metabolism. 67 (4): 317–23. doi:10.1006/mgme.1999.2879. PMID 10444342.
- ↑ Hassan FM, Khattab AA, Abo El Fotoh WM, Zidan RS (September 2017). "A66G and C524T polymorphisms of methionine synthase reductase gene are linked to the development of acyanotic congenital heart diseases in Egyptian children". Gene. 629: 59–63. doi:10.1016/j.gene.2017.07.081. PMID 28778621.
- ↑ G">"NM_002454.2(MTRR):c.1049A>G (p.Lys350Arg) AND not specified - ClinVar - NCBI". Retrieved 2017-09-16.
- ↑ G">"NM_002454.2(MTRR):c.1349C>G (p.Pro450Arg) AND Disorders of Intracellular Cobalamin Metabolism - ClinVar - NCBI". Retrieved 2017-09-16.
- ↑ Homolova K, Zavadakova P, Doktor TK, Schroeder LD, Kozich V, Andresen BS (April 2010). "The deep intronic c.903+469T>C mutation in the MTRR gene creates an SF2/ASF binding exonic splicing enhancer, which leads to pseudoexon activation and causes the cblE type of homocystinuria". Human Mutation. 31 (4): 437–44. doi:10.1002/humu.21206. PMC 3429857. PMID 20120036.
- ↑ T">"NM_002454.2(MTRR):c.1361C>T (p.Ser454Leu) Simple - Variation Report - ClinVar - NCBI". Retrieved 2017-09-16.
- ↑ A">"NM_002454.2(MTRR):c.1459G>A (p.Gly487Arg) AND Inborn genetic diseases - ClinVar - NCBI". Retrieved 2017-09-16.
- ↑ T">"NM_002454.2(MTRR):c.1573C>T (p.Arg525Ter) AND not provided - ClinVar - NCBI". Retrieved 2017-09-16.
- 1 2 3 4 5 6 Zavadáková P, Fowler B, Suormala T, Novotna Z, Mueller P, Hennermann JB, Zeman J, Vilaseca MA, Vilarinho L, Gutsche S, Wilichowski E, Horneff G, Kozich V (2005). "cblE type of homocystinuria due to methionine synthase reductase deficiency: functional correction by minigene expression". Human Mutation. 25 (3): 239–47. doi:10.1002/humu.20131. PMID 15714522. S2CID 17183484.
- ↑ A">"NM_002454.2(MTRR):c.1911G>A (p.Ala637=) Simple - Variation Report - ClinVar - NCBI". Retrieved 2017-09-16.
- 1 2 Wang P, Li S, Wang M, He J, Xi S (2017). "Association of MTRR A66G polymorphism with cancer susceptibility: Evidence from 85 studies". Journal of Cancer. 8 (2): 266–277. doi:10.7150/jca.17379. PMC 5327376. PMID 28243331.
- 1 2 3 4 Shi Q, Zhang Z, Li G, Pillow PC, Hernandez LM, Spitz MR, Wei Q (August 2005). "Polymorphisms of methionine synthase and methionine synthase reductase and risk of lung cancer: a case-control analysis". Pharmacogenetics and Genomics. 15 (8): 547–55. doi:10.1097/01.fpc.0000170916.96650.70. PMID 16006998. S2CID 25879819.
- ↑ Zhi X, Yang B, Fan S, Li Y, He M, Wang D, Wang Y, Wei J, Zheng Q, Sun G (December 2016). "Additive Interaction of MTHFR C677T and MTRR A66G Polymorphisms with Being Overweight/Obesity on the Risk of Type 2 Diabetes". International Journal of Environmental Research and Public Health. 13 (12): 1243. doi:10.3390/ijerph13121243. PMC 5201384. PMID 27983710.
- ↑ Matsuo K, Hamajima N, Hirai T, Kato T, Inoue M, Takezaki T, Tajima K (2002). "Methionine Synthase Reductase Gene A66G Polymorphism is Associated with Risk of Colorectal Cancer". Asian Pacific Journal of Cancer Prevention. 3 (4): 353–359. PMID 12716294.
- ↑ Sharp L, Little J (March 2004). "Polymorphisms in genes involved in folate metabolism and colorectal neoplasia: a HuGE review". American Journal of Epidemiology. 159 (5): 423–43. doi:10.1093/aje/kwh066. PMID 14977639.
- ↑ Wettergren Y, Odin E, Carlsson G, Gustavsson B (2010). "MTHFR, MTR, and MTRR polymorphisms in relation to p16INK4A hypermethylation in mucosa of patients with colorectal cancer". Molecular Medicine. 16 (9–10): 425–32. doi:10.2119/molmed.2009.00156. PMC 2935958. PMID 20549016.
- ↑ Le Marchand L, Donlon T, Hankin JH, Kolonel LN, Wilkens LR, Seifried A (2002). "B-vitamin intake, metabolic genes, and colorectal cancer risk (United States)". Cancer Causes & Control. 13 (3): 239–48. doi:10.1023/A:1015057614870. PMID 12020105. S2CID 6326507.
- ↑ Gemmati D, Ongaro A, Scapoli GL, Della Porta M, Tognazzo S, Serino ML, Di Bona E, Rodeghiero F, Gilli G, Reverberi R, Caruso A, Pasello M, Pellati A, De Mattei M (May 2004). "Common gene polymorphisms in the metabolic folate and methylation pathway and the risk of acute lymphoblastic leukemia and non-Hodgkin's lymphoma in adults". Cancer Epidemiology, Biomarkers & Prevention. 13 (5): 787–94. doi:10.1158/1055-9965.787.13.5. PMID 15159311. S2CID 14936869.
- ↑ Lin J, Spitz MR, Wang Y, Schabath MB, Gorlov IP, Hernandez LM, Pillow PC, Grossman HB, Wu X (September 2004). "Polymorphisms of folate metabolic genes and susceptibility to bladder cancer: a case-control study". Carcinogenesis. 25 (9): 1639–47. doi:10.1093/carcin/bgh175. PMID 15117811.
- ↑ Skibola CF, Forrest MS, Coppedé F, Agana L, Hubbard A, Smith MT, Bracci PM, Holly EA (October 2004). "Polymorphisms and haplotypes in folate-metabolizing genes and risk of non-Hodgkin lymphoma". Blood. 104 (7): 2155–62. doi:10.1182/blood-2004-02-0557. PMID 15198953.
- ↑ Stolzenberg-Solomon RZ, Qiao YL, Abnet CC, Ratnasinghe DL, Dawsey SM, Dong ZW, Taylor PR, Mark SD (November 2003). "Esophageal and gastric cardia cancer risk and folate- and vitamin B(12)-related polymorphisms in Linxian, China". Cancer Epidemiology, Biomarkers & Prevention. 12 (11 Pt 1): 1222–6. PMID 14652285.
- 1 2 3 Watkins D, Rosenblatt DS (June 1988). "Genetic heterogeneity among patients with methylcobalamin deficiency. Definition of two complementation groups, cblE and cblG". The Journal of Clinical Investigation. 81 (6): 1690–4. doi:10.1172/JCI113507. PMC 442612. PMID 3384945.
- 1 2 3 4 5 6 7 8 9 Zavadakova P, Fowler B, Zeman J, Suormala T, Pristoupilová K, Kozich V, Zavad'áková P (October 2002). "CblE type of homocystinuria due to methionine synthase reductase deficiency: clinical and molecular studies and prenatal diagnosis in two families". Journal of Inherited Metabolic Disease. 25 (6): 461–76. doi:10.1023/A:1021299117308. PMID 12555939. S2CID 9114486.
- 1 2 3 4 5 6 7 Botto N, Andreassi MG, Manfredi S, Masetti S, Cocci F, Colombo MG, Storti S, Rizza A, Biagini A (September 2003). "Genetic polymorphisms in folate and homocysteine metabolism as risk factors for DNA damage". European Journal of Human Genetics. 11 (9): 671–8. doi:10.1038/sj.ejhg.5201024. PMID 12939653.
- 1 2 Brown CA, McKinney KQ, Kaufman JS, Gravel RA, Rozen R (June 2000). "A common polymorphism in methionine synthase reductase increases risk of premature coronary artery disease". Journal of Cardiovascular Risk. 7 (3): 197–200. doi:10.1177/204748730000700306. PMID 11006889. S2CID 32898984.
- ↑ Shaw GM, Lu W, Zhu H, Yang W, Briggs FB, Carmichael SL, Barcellos LF, Lammer EJ, Finnell RH (June 2009). "118 SNPs of folate-related genes and risks of spina bifida and conotruncal heart defects". BMC Medical Genetics. 10: 49. doi:10.1186/1471-2350-10-49. PMC 2700092. PMID 19493349.
- ↑ Balduino Victorino D, de Godoy MF, Goloni-Bertollo EM, Pavarino ÉC (2014). "Genetic polymorphisms involved in folate metabolism and maternal risk for down syndrome: a meta-analysis". Disease Markers. 2014: 1–12. doi:10.1155/2014/517504. PMC 4269293. PMID 25544792.
- ↑ Muthuswamy S, Agarwal S (2016). "Do the MTHFR gene polymorphism and Down syndrome pregnancy association stands true? A case–control study of Indian population and meta-analysis". Egyptian Journal of Medical Human Genetics. 17 (1): 87–97. doi:10.1016/j.ejmhg.2015.08.003. ISSN 1110-8630.
- 1 2 3 O'Leary VB, Parle-McDermott A, Molloy AM, Kirke PN, Johnson Z, Conley M, Scott JM, Mills JL (January 2002). "MTRR and MTHFR polymorphism: link to Down syndrome?". American Journal of Medical Genetics. 107 (2): 151–5. doi:10.1002/ajmg.10121. PMID 11807890.
- 1 2 Martínez-Frías ML, Pérez B, Desviat LR, Castro M, Leal F, Rodríguez L, Mansilla E, Martínez-Fernández ML, Bermejo E, Rodríguez-Pinilla E, Prieto D, Ugarte M (May 2006). "Maternal polymorphisms 677C-T and 1298A-C of MTHFR, and 66A-G MTRR genes: is there any relationship between polymorphisms of the folate pathway, maternal homocysteine levels, and the risk for having a child with Down syndrome?". American Journal of Medical Genetics. Part A. 140 (9): 987–97. doi:10.1002/ajmg.a.31203. PMID 16575899. S2CID 44819166.
- ↑ Bosco P, Guéant-Rodriguez RM, Anello G, Barone C, Namour F, Caraci F, Romano A, Romano C, Guéant JL (September 2003). "Methionine synthase (MTR) 2756 (A --> G) polymorphism, double heterozygosity methionine synthase 2756 AG/methionine synthase reductase (MTRR) 66 AG, and elevated homocysteinemia are three risk factors for having a child with Down syndrome". American Journal of Medical Genetics. Part A. 121A (3): 219–24. doi:10.1002/ajmg.a.20234. PMID 12923861. S2CID 38425348.
- ↑ Al-Gazali LI, Padmanabhan R, Melnyk S, Yi P, Pogribny IP, Pogribna M, Bakir M, Hamid ZA, Abdulrazzaq Y, Dawodu A, James SJ (October 2001). "Abnormal folate metabolism and genetic polymorphism of the folate pathway in a child with Down syndrome and neural tube defect". American Journal of Medical Genetics. 103 (2): 128–32. doi:10.1002/ajmg.1509. PMID 11568918.
Further reading
- Wilson A, Platt R, Wu Q, Leclerc D, Christensen B, Yang H, Gravel RA, Rozen R (August 1999). "A common variant in methionine synthase reductase combined with low cobalamin (vitamin B12) increases risk for spina bifida". Molecular Genetics and Metabolism. 67 (4): 317–23. doi:10.1006/mgme.1999.2879. PMID 10444342.
- Wilson A, Leclerc D, Rosenblatt DS, Gravel RA (October 1999). "Molecular basis for methionine synthase reductase deficiency in patients belonging to the cblE complementation group of disorders in folate/cobalamin metabolism". Human Molecular Genetics. 8 (11): 2009–16. doi:10.1093/hmg/8.11.2009. PMID 10484769.
- James SJ, Pogribna M, Pogribny IP, Melnyk S, Hine RJ, Gibson JB, Yi P, Tafoya DL, Swenson DH, Wilson VL, Gaylor DW (October 1999). "Abnormal folate metabolism and mutation in the methylenetetrahydrofolate reductase gene may be maternal risk factors for Down syndrome". The American Journal of Clinical Nutrition. 70 (4): 495–501. doi:10.1093/ajcn/70.4.495. PMID 10500018.
- Leclerc D, Odièvre M, Wu Q, Wilson A, Huizenga JJ, Rozen R, Scherer SW, Gravel RA (November 1999). "Molecular cloning, expression and physical mapping of the human methionine synthase reductase gene". Gene. 240 (1): 75–88. doi:10.1016/S0378-1119(99)00431-X. PMID 10564814.
- Doolin MT, Barbaux S, McDonnell M, Hoess K, Whitehead AS, Mitchell LE (November 2002). "Maternal genetic effects, exerted by genes involved in homocysteine remethylation, influence the risk of spina bifida". American Journal of Human Genetics. 71 (5): 1222–6. doi:10.1086/344209. PMC 385102. PMID 12375236.
- Olteanu H, Munson T, Banerjee R (November 2002). "Differences in the efficiency of reductive activation of methionine synthase and exogenous electron acceptors between the common polymorphic variants of human methionine synthase reductase". Biochemistry. 41 (45): 13378–85. doi:10.1021/bi020536s. PMID 12416982.
- Zavadakova P, Fowler B, Zeman J, Suormala T, Pristoupilová K, Kozich V, Zavad'áková P (October 2002). "CblE type of homocystinuria due to methionine synthase reductase deficiency: clinical and molecular studies and prenatal diagnosis in two families". Journal of Inherited Metabolic Disease. 25 (6): 461–76. doi:10.1023/A:1021299117308. PMID 12555939. S2CID 9114486.
- Pietrzyk JJ, Bik-Multanowski M, Sanak M, Twardowska M (2003). "Polymorphisms of the 5,10-methylenetetrahydrofolate and the methionine synthase reductase genes as independent risk factors for spina bifida". Journal of Applied Genetics. 44 (1): 111–3. PMID 12590188.
- Zhu H, Wicker NJ, Shaw GM, Lammer EJ, Hendricks K, Suarez L, Canfield M, Finnell RH (March 2003). "Homocysteine remethylation enzyme polymorphisms and increased risks for neural tube defects". Molecular Genetics and Metabolism. 78 (3): 216–21. doi:10.1016/S1096-7192(03)00008-8. PMID 12649067.
- Brilakis ES, Berger PB, Ballman KV, Rozen R (June 2003). "Methylenetetrahydrofolate reductase (MTHFR) 677C>T and methionine synthase reductase (MTRR) 66A>G polymorphisms: association with serum homocysteine and angiographic coronary artery disease in the era of flour products fortified with folic acid". Atherosclerosis. 168 (2): 315–22. doi:10.1016/S0021-9150(03)00098-4. PMID 12801615.
- Sliwerska E, Szpecht-Potocka A (2003). "[Mutations of MTHFR, MTR, MTRR genes as high risk factors for neural tube defects]". Medycyna Wieku Rozwojowego. 6 (4): 371–82. PMID 12810988.
- Beyer K, Lao JI, Latorre P, Riutort N, Matute B, Fernández-Figueras MT, Mate JL, Ariza A (July 2003). "Methionine synthase polymorphism is a risk factor for Alzheimer disease". NeuroReport. 14 (10): 1391–4. doi:10.1097/01.wnr.0000073683.00308.0e. PMID 12876480. S2CID 33245738.
- Bosco P, Guéant-Rodriguez RM, Anello G, Barone C, Namour F, Caraci F, Romano A, Romano C, Guéant JL (September 2003). "Methionine synthase (MTR) 2756 (A --> G) polymorphism, double heterozygosity methionine synthase 2756 AG/methionine synthase reductase (MTRR) 66 AG, and elevated homocysteinemia are three risk factors for having a child with Down syndrome". American Journal of Medical Genetics. Part A. 121A (3): 219–24. doi:10.1002/ajmg.a.20234. PMID 12923861. S2CID 38425348.
- Olteanu H, Wolthers KR, Munro AW, Scrutton NS, Banerjee R (February 2004). "Kinetic and thermodynamic characterization of the common polymorphic variants of human methionine synthase reductase". Biochemistry. 43 (7): 1988–97. doi:10.1021/bi035910i. PMID 14967039.
- Gemmati D, Ongaro A, Scapoli GL, Della Porta M, Tognazzo S, Serino ML, Di Bona E, Rodeghiero F, Gilli G, Reverberi R, Caruso A, Pasello M, Pellati A, De Mattei M (May 2004). "Common gene polymorphisms in the metabolic folate and methylation pathway and the risk of acute lymphoblastic leukemia and non-Hodgkin's lymphoma in adults". Cancer Epidemiology, Biomarkers & Prevention. 13 (5): 787–94. doi:10.1158/1055-9965.787.13.5. PMID 15159311. S2CID 14936869.
- Leal NA, Olteanu H, Banerjee R, Bobik TA (November 2004). "Human ATP:Cob(I)alamin adenosyltransferase and its interaction with methionine synthase reductase". The Journal of Biological Chemistry. 279 (46): 47536–42. doi:10.1074/jbc.M405449200. PMID 15347655.
- Vaughn JD, Bailey LB, Shelnutt KP, Dunwoody KM, Maneval DR, Davis SR, Quinlivan EP, Gregory JF, Theriaque DW, Kauwell GP (November 2004). "Methionine synthase reductase 66A->G polymorphism is associated with increased plasma homocysteine concentration when combined with the homozygous methylenetetrahydrofolate reductase 677C->T variant". The Journal of Nutrition. 134 (11): 2985–90. doi:10.1093/jn/134.11.2985. PMID 15514263.