 | |
| Names | |
|---|---|
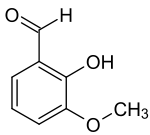
| Preferred IUPAC name
2-Hydroxy-3-methoxybenzaldehyde | |
| Other names
o-Vanillin 3-Methoxysalicylaldehyde | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.197 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H8O3 | |
| Molar mass | 152.15 g/mol |
| Appearance | Yellow, fibrous solid |
| Density | 1.231 g/mL |
| Melting point | 40 to 42 °C (104 to 108 °F; 313 to 315 K) |
| Boiling point | 265 to 266 °C (509 to 511 °F; 538 to 539 K) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
May cause irritation to skin, eyes, and respiratory tract |
| GHS labelling: | |
  | |
| Danger | |
| H302, H315, H317, H318, H319, H335 | |
| P261, P264, P270, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P330, P332+P313, P333+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
| Flash point | > 110 °C (230 °F; 383 K) |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Related compounds |
Eugenol, Anisaldehyde, Phenol, Vanillin |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
ortho-Vanillin (2-hydroxy-3-methoxybenzaldehyde) is an organic solid present in the extracts and essential oils of many plants.[1][2][3] Its functional groups include aldehyde, ether and phenol. ortho-Vanillin, a compound of the formula C8H8O3, is distinctly different from its more prevalent isomer, meta-vanillin. The "ortho-" prefix refers to the position of the compound’s hydroxyl moiety, which is found in the para-position in vanillin.
ortho-Vanillin is a fibrous, light-yellow, crystalline solid. Present in a variety of food products, it is not specifically sought after, and is therefore a less-commonly produced and encountered food additive.
History
ortho-Vanillin was first isolated, in 1876, by renowned German chemist Ferdinand Tiemann.[4] By 1910, methods for its purification had been developed by Francis Noelting, who similarly demonstrated its versatility as a general synthetic precursor for a diverse array of compounds, such as the coumarins.[5]
By 1920, the compound began to show use as a dye for hides.[6]
Biological properties
ortho-Vanillin is harmful if ingested, irritating to eyes, skin and respiratory system, but has an unmistakable high LD50 of 1330 mg/kg in mice.[7]
It is a weak inhibitor of tyrosinase,[8] and displays both antimutagenic and comutagenic properties in Escherichia coli.[9] However, its net effect makes it a “potent comutagen”.[10]
ortho-Vanillin possesses moderate antifungal and antibacterial properties.[11]
Uses
Today, most ortho-vanillin is used in the study of mutagenesis and as a synthetic precursor for pharmaceuticals, for example, benafentrine[12] and an antiandrogen compound called Pentomone.
See also
Notes
- ↑ Abou Zeid, A. H.; Sleem, A. A. (2002). "Natural and stress constituents from Spinacia oleracea L. leaves and their biological activities". Bulletin of the Faculty of Pharmacy (Cairo University). 40 (2): 153–167.
- ↑ Barbe, Jean-Christophe; Bertrand, Alain. (1996). "Quantitative analysis of volatile compounds stemming from oak wood. Application to the aging of wines in barrels". Journal des Sciences et Techniques de la Tonnellerie. 2: 77–88.
- ↑ Brunke, E. J.; Hammerschmidt, F. J.; Schmaus, G. (1992). "Das etherische Öl von Santolina chamaecyparissus L." [The essential oil of Santolina chamaecyparissus L.]. Parfümerie und Kosmetik. 73 (9): 617–637.
- ↑ Tiemann, Ferdinand (1876). "Ueber die der Coniferyl- und Vanillinreihe angehörigen Verbindungen" [On coniferyl and vanillin series-related compounds]. Berichte der Deutschen Chemischen Gesellschaft. 9: 409–423. doi:10.1002/cber.187600901133.
- ↑ Noelting, Francis A. M. (1910). "o-Hydroxy-m-methoxybenzaldehyde (Orthovanillin)". Annales de Chimie et de Physique. 19: 476–550.
- ↑ Gerngross, Otto (1920). "Dyeing hide with o-vanillin and o-protocatechualdehyde and aldehyde tanning". Angewandte Chemie. 33 (44): 136–138. doi:10.1002/ange.19200334403.
- ↑ http://msds.chem.ox.ac.uk/VA/o-vanillin.html
- ↑ Kubo, Isao; Kinst-Hori, Ikuyo (1999). "Tyrosinase inhibitory activity of the olive oil flavor compounds". Journal of Agricultural and Food Chemistry. 47 (11): 4574–4578. doi:10.1021/jf990165v. PMID 10552852.
- ↑ Watanabe, Kazuko; Ohta, Toshihiro; Shirasu, Yasuhiko (1989). "Enhancement and inhibition of mutation by o-vanillin in Escherichia coli". Mutation Research/DNA Repair. 218 (2): 105–9. doi:10.1016/0921-8777(89)90016-5. PMID 2671704.
- ↑ Takahashi, Kazuhiko; Sekiguchi, Mutsuo; Kawazoe, Yutaka (1989). "A specific inhibition of induction of adaptive response by o-vanillin, a potent comutagen". Biochemical and Biophysical Research Communications. 162 (3): 1376–81. doi:10.1016/0006-291X(89)90826-7. PMID 2669748.
- ↑ Leifertova, I.; Hejtmankova, N.; Hlava, H.; Kudrnacova, J.; Santavy, F. (1975). "Antifungal and antibacterial effects of phenolic substances. A study of the relation between the biological activity and the constitution of the investigated compounds". Acta Universitatis Palackianae Olomucensis, Facultatis Medicae. 74: 83–101.
- ↑ Press, Jeffery B.; Bandurco, Victor T.; Wong, Elizabeth M.; Hajos, Zoltan G.; Kanojia, Ramesh M.; Mallory, Robert A.; Deegan, Edward G.; Mcnally, James J.; Roberts, Jerry R.; Cotter, Mary Lou; Graden, David W.; Lloyd, John R. (1986). "Synthesis of 5,6-dimethoxyquinazolin-2(1H)-ones". Journal of Heterocyclic Chemistry. 23 (6): 1821–1828. doi:10.1002/jhet.5570230643. ISSN 0022-152X.