 | |
| Names | |
|---|---|
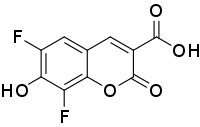
| Preferred IUPAC name
6,8-Difluoro-7-hydroxy-2-oxo-2H-1-benzopyran-3-carboxylic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C10H4F2O7 | |
| Molar mass | 274.132 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Pacific Blue, or systematically 3-carboxy-6,8-difluoro-7-hydroxycoumarin, is a fluorophore used in cell biology.[1] Its excitation maximum lies at 401 nm, while its emission maximum is at 452 nm.[2] In contrast to the less acidic 7-hydroxy-3-carboxycoumarin (pKa=7.0), the high acidity of the phenol of Pacific Blue (pKa=3.7) causes its fluorescence to remain very high at neutral pH.
Pacific Blue is a member of the group of Pacific dyes, which includes Pacific Orange, Pacific Green, and Pacific Blue. These fluorescent dyes all have an absorption maximum between 400 and 410 nm, but with different emission spectra: this allows simultaneous excitation with one laser, producing emission at maxima of 551nm, 500 nm and 455 nm, respectively[3] (note: absorption and emission maxima may vary depending on the manufacturer).
In flow cytometry Pacific Blue (as well as other Pacific dyes) absorbs near 405 nm, disallowing simultaneous use with other fluorophores which share similar excitation and/or emission spectra, such as Brilliant Violet 421 (BV421) or V450.[4]
Usage
For purposes of immunohistochemistry or targeted staining in general, reactive forms of Pacific Blue are used to label targeting antibodies. Derivatives of Pacific Blue have been used to study small molecule-protein interactions using fluorescence resonance energy transfer (FRET).[5] Pacific Blue has also been used to study interactions of the anticancer drug paclitaxel (Taxol) with tubulin in living cells.[6] Reactive forms in use include Pacific Blue succinimidyl ester and Pacific Blue C5-Maleimide.[3]
References
- ↑ Lee, Wei-Chuan (1998). "Synthesis of Novel Fluorinated Coumarins: Excellent UV-Light Excitable Fluorescent Dyes". Bioorganic & Medicinal Chemistry Letters. 8 (1): 3107–3110. doi:10.1016/s0960-894x(98)00578-2. PMID 9873685.
- ↑ "Absorption and Emission Spectra — Pacific Blue™". BD Biosciences.
- 1 2 "Pacific Blue dye". ThermoFisher Scientific.
- ↑ "Violet Proliferation Dye 450". BD Biosciences.
- ↑ Lee, Molly M. (2016). "Quantification of Small Molecule–Protein Interactions using FRET between Tryptophan and the Pacific Blue Fluorophore". ACS Omega. 6 (1): 1266–1276. doi:10.1021/acsomega.6b00356. PMC 5204206. PMID 28058293.
- ↑ Lee, Molly M. (2017). "Synthesis of a Fluorescent Analogue of Paclitaxel that Selectively Binds Microtubules and Sensitively Detects Efflux by P-Glycoprotein". Angewandte Chemie International Edition. 56 (1): 6927–6931. doi:10.1002/anie.201703298. PMC 5679005. PMID 28485901.