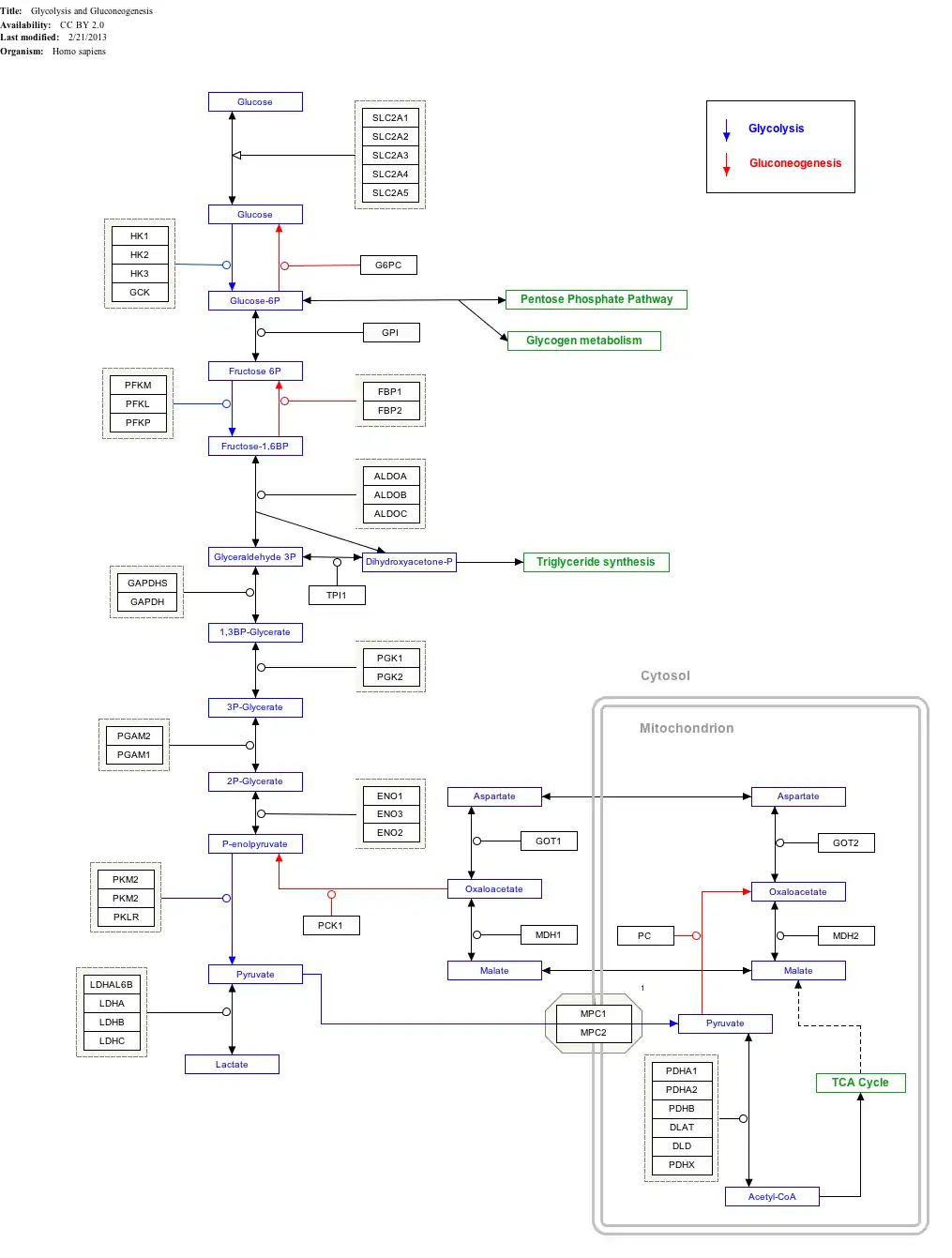
| Phosphoglycerate kinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 | |||||||||
| Identifiers | |||||||||
| EC no. | 2.7.2.3 | ||||||||
| CAS no. | 9001-83-6 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Phosphoglycerate kinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
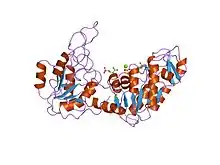 Structure of yeast phosphoglycerate kinase.[1] | |||||||||
| Identifiers | |||||||||
| Symbol | PGK | ||||||||
| Pfam | PF00162 | ||||||||
| InterPro | IPR001576 | ||||||||
| PROSITE | PDOC00102 | ||||||||
| SCOP2 | 3pgk / SCOPe / SUPFAM | ||||||||
| |||||||||
Phosphoglycerate kinase (EC 2.7.2.3) (PGK 1) is an enzyme that catalyzes the reversible transfer of a phosphate group from 1,3-bisphosphoglycerate (1,3-BPG) to ADP producing 3-phosphoglycerate (3-PG) and ATP :
- 1,3-bisphosphoglycerate + ADP ⇌ glycerate 3-phosphate + ATP
Like all kinases it is a transferase. PGK is a major enzyme used in glycolysis, in the first ATP-generating step of the glycolytic pathway. In gluconeogenesis, the reaction catalyzed by PGK proceeds in the opposite direction, generating ADP and 1,3-BPG.
In humans, two isozymes of PGK have been so far identified, PGK1 and PGK2. The isozymes have 87-88% identical amino acid sequence identity and though they are structurally and functionally similar, they have different localizations: PGK2, encoded by an autosomal gene, is unique to meiotic and postmeiotic spermatogenic cells, while PGK1, encoded on the X-chromosome, is ubiquitously expressed in all cells.[2]
Biological function
PGK is present in all living organisms as one of the two ATP-generating enzymes in glycolysis. In the gluconeogenic pathway, PGK catalyzes the reverse reaction. Under biochemical standard conditions, the glycolytic direction is favored.[1]
In the Calvin cycle in photosynthetic organisms, PGK catalyzes the phosphorylation of 3-PG, producing 1,3-BPG and ADP, as part of the reactions that regenerate ribulose-1,5-bisphosphate.
PGK has been reported to exhibit thiol reductase activity on plasmin, leading to angiostatin formation, which inhibits angiogenesis and tumor growth. The enzyme was also shown to participate in DNA replication and repair in mammal cell nuclei.[3]
The human isozyme PGK2, which is only expressed during spermatogenesis, was shown to be essential for sperm function in mice.[4]
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
- ↑ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
Structure
Overview
PGK is found in all living organisms and its sequence has been highly conserved throughout evolution. The enzyme exists as a 415-residue monomer containing two nearly equal-sized domains that correspond to the N- and C-termini of the protein.[5] 3-phosphoglycerate (3-PG) binds to the N-terminal, while the nucleotide substrates, MgATP or MgADP, bind to the C-terminal domain of the enzyme. This extended two-domain structure is associated with large-scale 'hinge-bending' conformational changes, similar to those found in hexokinase.[6] The two domains of the protein are separated by a cleft and linked by two alpha-helices.[2] At the core of each domain is a 6-stranded parallel beta-sheet surrounded by alpha helices. The two lobes are capable of folding independently, consistent with the presence of intermediates on the folding pathway with a single domain folded.[7][8] Though the binding of either substrate triggers a conformational change, only through the binding of both substrates does domain closure occur, leading to the transfer of the phosphate group.[2]
The enzyme has a tendency to exist in the open conformation with short periods of closure and catalysis, which allow for rapid diffusion of substrate and products through the binding sites; the open conformation of PGK is more conformationally stable due to the exposure of a hydrophobic region of the protein upon domain closure.[7]
Role of magnesium
Magnesium ions are normally complexed to the phosphate groups the nucleotide substrates of PGK. It is known that in the absence of magnesium, no enzyme activity occurs.[9] The bivalent metal assists the enzyme ligands in shielding the bound phosphate group's negative charges, allowing the nucleophilic attack to occur; this charge-stabilization is a typical characteristic of phosphotransfer reaction.[10] It is theorized that the ion may also encourage domain closure when PGK has bound both substrates.[9]
Mechanism
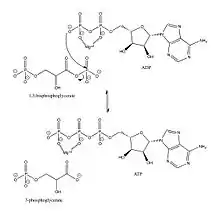
Without either substrate bound, PGK exists in an "open" conformation. After both the triose and nucleotide substrates are bound to the N- and C-terminal domains, respectively, an extensive hinge-bending motion occurs, bringing the domains and their bound substrates into close proximity and leading to a "closed" conformation.[11] Then, in the case of the forward glycolytic reaction, the beta-phosphate of ADP initiates a nucleophilic attack on the 1-phosphate of 1,3-BPG. The Lys219 on the enzyme guides the phosphate group to the substrate.
PGK proceeds through a charge-stabilized transition state that is favored over the arrangement of the bound substrate in the closed enzyme because in the transition state, all three phosphate oxygens are stabilized by ligands, as opposed to only two stabilized oxygens in the initial bound state.[12]
In the glycolytic pathway, 1,3-BPG is the phosphate donor and has a high phosphoryl-transfer potential. The PGK-catalyzed transfer of the phosphate group from 1,3-BPG to ADP to yield ATP can power the carbon-oxidation reaction of the previous glycolytic step (converting glyceraldehyde 3-phosphate to 3-phosphoglycerate).
Regulation
The enzyme is activated by low concentrations of various multivalent anions, such as pyrophosphate, sulfate, phosphate, and citrate. High concentrations of MgATP and 3-PG activates PGK, while Mg2+ at high concentrations non-competitively inhibits the enzyme.[13]
PGK exhibits a wide specificity toward nucleotide substrates.[14] Its activity is inhibited by salicylates, which appear to mimic the enzyme's nucleotide substrate.[15]
Macromolecular crowding has been shown to increase PGK activity in both computer simulations and in vitro environments simulating a cell interior; as a result of crowding, the enzyme becomes more enzymatically active and more compact.[5]
Disease relevance
Phosphoglycerate kinase (PGK) deficiency is an X-linked recessive trait associated with hemolytic anemia, mental disorders and myopathy in humans,[16][17] depending on form – there exists a hemolytic form and a myopathic form.[18] Since the trait is X-linked, it is usually fully expressed in males, who have one X chromosome; affected females are typically asymptomatic.[2][17] The condition results from mutations in Pgk1, the gene encoding PGK1, and twenty mutations have been identified.[17][2] On a molecular level, the mutation in Pgk1 impairs the thermal stability and inhibits the catalytic activity of the enzyme.[2] PGK is the only enzyme in the immediate glycolytic pathway encoded by an X-linked gene. In the case of hemolytic anemia, PGK deficiency occurs in the erythrocytes. Currently, no definitive treatment exists for PGK deficiency.[19]
PGK1 overexpression has been associated with gastric cancer and has been found to increase the invasiveness of gastric cancer cells in vitro.[20] The enzyme is secreted by tumor cells and participates in the angiogenic process, leading to the release of angiostatin and the inhibition of tumor blood vessel growth.[3]
Due to its wide specificity towards nucleotide substrates, PGK is known to participate in the phosphorylation and activation of HIV antiretroviral drugs, which are nucleotide-based.[14][21]
Human isozymes
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
- 1 2 Watson HC, Walker NP, Shaw PJ, Bryant TN, Wendell PL, Fothergill LA, Perkins RE, Conroy SC, Dobson MJ, Tuite MF (1982). "Sequence and structure of yeast phosphoglycerate kinase". The EMBO Journal. 1 (12): 1635–40. doi:10.1002/j.1460-2075.1982.tb01366.x. PMC 553262. PMID 6765200.
- 1 2 3 4 5 6 Chiarelli LR, Morera SM, Bianchi P, Fermo E, Zanella A, Galizzi A, Valentini G (2012). "Molecular insights on pathogenic effects of mutations causing phosphoglycerate kinase deficiency". PLOS ONE. 7 (2): e32065. Bibcode:2012PLoSO...732065C. doi:10.1371/journal.pone.0032065. PMC 3279470. PMID 22348148.
- 1 2 Lay AJ, Jiang XM, Kisker O, Flynn E, Underwood A, Condron R, Hogg PJ (December 2000). "Phosphoglycerate kinase acts in tumour angiogenesis as a disulphide reductase". Nature. 408 (6814): 869–73. Bibcode:2000Natur.408..869L. doi:10.1038/35048596. PMID 11130727. S2CID 4340557.
- ↑ Danshina PV, Geyer CB, Dai Q, Goulding EH, Willis WD, Kitto GB, McCarrey JR, Eddy EM, O'Brien DA (January 2010). "Phosphoglycerate kinase 2 (PGK2) is essential for sperm function and male fertility in mice". Biology of Reproduction. 82 (1): 136–45. doi:10.1095/biolreprod.109.079699. PMC 2802118. PMID 19759366.
- 1 2 Dhar A, Samiotakis A, Ebbinghaus S, Nienhaus L, Homouz D, Gruebele M, Cheung MS (October 2010). "Structure, function, and folding of phosphoglycerate kinase are strongly perturbed by macromolecular crowding". Proceedings of the National Academy of Sciences of the United States of America. 107 (41): 17586–91. Bibcode:2010PNAS..10717586D. doi:10.1073/pnas.1006760107. PMC 2955104. PMID 20921368.
- ↑ Kumar S, Ma B, Tsai CJ, Wolfson H, Nussinov R (1999). "Folding funnels and conformational transitions via hinge-bending motions". Cell Biochemistry and Biophysics. 31 (2): 141–64. doi:10.1007/BF02738169. PMID 10593256. S2CID 41924983.
- 1 2 Yon JM, Desmadril M, Betton JM, Minard P, Ballery N, Missiakas D, Gaillard-Miran S, Perahia D, Mouawad L (1990). "Flexibility and folding of phosphoglycerate kinase". Biochimie. 72 (6–7): 417–29. doi:10.1016/0300-9084(90)90066-p. PMID 2124145.
- ↑ Zerrad L, Merli A, Schröder GF, Varga A, Gráczer É, Pernot P, Round A, Vas M, Bowler MW (April 2011). "A spring-loaded release mechanism regulates domain movement and catalysis in phosphoglycerate kinase". The Journal of Biological Chemistry. 286 (16): 14040–8. doi:10.1074/jbc.M110.206813. PMC 3077604. PMID 21349853.
- 1 2 Varga A, Palmai Z, Gugolya Z, Gráczer É, Vonderviszt F, Závodszky P, Balog E, Vas M (December 2012). "Importance of aspartate residues in balancing the flexibility and fine-tuning the catalysis of human 3-phosphoglycerate kinase". Biochemistry. 51 (51): 10197–207. doi:10.1021/bi301194t. PMID 23231058.
- ↑ Cliff MJ, Bowler MW, Varga A, Marston JP, Szabó J, Hounslow AM, Baxter NJ, Blackburn GM, Vas M, Waltho JP (May 2010). "Transition state analogue structures of human phosphoglycerate kinase establish the importance of charge balance in catalysis". Journal of the American Chemical Society. 132 (18): 6507–16. doi:10.1021/ja100974t. PMID 20397725.
- ↑ Banks, R. D.; Blake, C. C. F.; Evans, P. R.; Haser, R.; Rice, D. W.; Hardy, G. W.; Merrett, M.; Phillips, A. W. (28 June 1979). "Sequence, structure and activity of phosphoglycerate kinase: a possible hinge-bending enzyme". Nature. 279 (5716): 773–777. Bibcode:1979Natur.279..773B. doi:10.1038/279773a0. PMID 450128. S2CID 4321999.
- ↑ Bernstein BE, Hol WG (March 1998). "Crystal structures of substrates and products bound to the phosphoglycerate kinase active site reveal the catalytic mechanism". Biochemistry. 37 (13): 4429–36. doi:10.1021/bi9724117. PMID 9521762.
- ↑ Larsson-Raźnikiewicz M (January 1967). "Kinetic studies on the reaction catalyzed by phosphoglycerate kinase. II. The kinetic relationships between 3-phosphoglycerate, MgATP2-and activating metal ion". Biochimica et Biophysica Acta (BBA) - Enzymology. 132 (1): 33–40. doi:10.1016/0005-2744(67)90189-1. PMID 6030358.
- 1 2 Varga A, Chaloin L, Sági G, Sendula R, Gráczer E, Liliom K, Závodszky P, Lionne C, Vas M (June 2011). "Nucleotide promiscuity of 3-phosphoglycerate kinase is in focus: implications for the design of better anti-HIV analogues". Molecular BioSystems. 7 (6): 1863–73. doi:10.1039/c1mb05051f. PMID 21505655.
- ↑ Larsson-Raźnikiewicz, Märtha; Wiksell, Eva (1 March 1978). "Inhibition of phosphoglycerate kinase by salicylates". Biochimica et Biophysica Acta (BBA) - Enzymology. 523 (1): 94–100. doi:10.1016/0005-2744(78)90012-8. PMID 343818.
- ↑ Yoshida A, Tani K (1983). "Phosphoglycerate kinase abnormalities: functional, structural and genomic aspects". Biomedica Biochimica Acta. 42 (11–12): S263-7. PMID 6689547.
- 1 2 3 Beutler E (January 2007). "PGK deficiency". British Journal of Haematology. 136 (1): 3–11. doi:10.1111/j.1365-2141.2006.06351.x. PMID 17222195. S2CID 21111736.
- ↑ NIH Genetics Home Reference
- ↑ Rhodes M, Ashford L, Manes B, Calder C, Domm J, Frangoul H (February 2011). "Bone marrow transplantation in phosphoglycerate kinase (PGK) deficiency". British Journal of Haematology. 152 (4): 500–2. doi:10.1111/j.1365-2141.2010.08474.x. PMID 21223252. S2CID 37605904.
- ↑ Zieker D, Königsrainer I, Tritschler I, Löffler M, Beckert S, Traub F, Nieselt K, Bühler S, Weller M, Gaedcke J, Taichman RS, Northoff H, Brücher BL, Königsrainer A (March 2010). "Phosphoglycerate kinase 1 a promoting enzyme for peritoneal dissemination in gastric cancer". International Journal of Cancer. 126 (6): 1513–20. doi:10.1002/ijc.24835. PMC 2811232. PMID 19688824.
- ↑ Gallois-Montbrun S, Faraj A, Seclaman E, Sommadossi JP, Deville-Bonne D, Véron M (November 2004). "Broad specificity of human phosphoglycerate kinase for antiviral nucleoside analogs". Biochemical Pharmacology. 68 (9): 1749–56. doi:10.1016/j.bcp.2004.06.012. PMID 15450940.
External links
- Phosphoglycerate+kinase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Illustration at arizona.edu