 | |
| Names | |
|---|---|
| IUPAC name
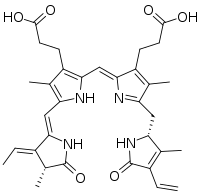
3-[(2Z,5Z)-2-[[3-(2-carboxyethyl)-5-[[(2R)-4-ethenyl-3-methyl-5-oxo-1,2-dihydropyrrol-2-yl]methyl]-4-methyl-1H-pyrrol-2-yl]methylidene]-5-[[(3E,4R)-3-ethylidene-4-methyl-5-oxopyrrol-2-yl]methylidene]-4-methylpyrrol-3-yl]propanoic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| MeSH | phycoerythrobilin |
PubChem CID |
|
| |
| |
| Properties | |
| C33H38N4O6 | |
| Molar mass | 586.689 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Phycoerythrobilin is a red phycobilin, i.e. an open tetrapyrrole chromophore[1] found in cyanobacteria and in the chloroplasts of red algae, glaucophytes and some cryptomonads. Phycoerythrobilin is present in the phycobiliprotein phycoerythrin, of which it is the terminal acceptor of energy. The amount of phycoerythrobilin in phycoerythrins varies a lot, depending on the considered organism. In some Rhodophytes and oceanic cyanobacteria, phycoerythrobilin is also present in the phycocyanin, then termed R-phycocyanin. Like all phycobilins, phycoerythrobilin is covalently linked to these phycobiliproteins by a thioether bond.
References
- ↑ Chapman, David J.; Cole, W. J.; Siegelman, Harold W. (1967). "Structure of phycoerythrobilin". Journal of the American Chemical Society. 89 (23): 5976–5977. doi:10.1021/ja00999a058. ISSN 0002-7863.
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.