 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Phenylpropan-1-one | |
| Other names
Ethyl phenyl ketone, BzEt | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.053 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H10O | |
| Molar mass | 134.178 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.0087 g/mL |
| Melting point | 18.6 °C (65.5 °F; 291.8 K) |
| Boiling point | 218 °C (424 °F; 491 K) |
| Insoluble | |
| -83.73·10−6 cm3/mol | |
| Related compounds | |
Related ketones |
Acetophenone Butyrophenone |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
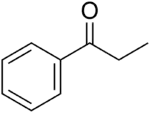
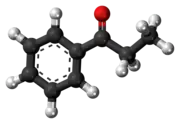
Propiophenone (shorthand: benzoylethane or BzEt) is an aryl ketone. It is a colorless, sweet-smelling liquid that is insoluble in water, but miscible with organic solvents. It is used in the preparation of other compounds.
Production
Propiophenone can be prepared by Friedel–Crafts reaction of propanoyl chloride and benzene. It is also prepared commercially by ketonization of benzoic acid and propionic acid over calcium acetate and alumina at 450–550 °C:[1]
- C6H5CO2H + CH3CH2CO2H → C6H5C(O)CH2CH3 + CO2 + H2O
Ludwig Claisen discovered that α-methoxystyrene forms this compound when heated for an hour at 300 °C (65% yield).[2][3]
Uses
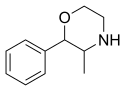
It is an intermediate in the synthesis of the pharmaceuticals phenmetrazine and propoxyphen.[1][4][5]
Other drugs made from propiophenone include the following: PDM-35, Eprazinone, Methcathinone (leading to ephedrine), Trimebutine, Amfepramone, Diphepanol, Metamfepramone, Etoxadrol, Hydroxyphenamate, Phendimetrazine, Iminophenimide, Bencisteine, Flumecinol, Pyrroliphene, Perisone,
References
- 1 2 Siegel, H.; Eggersdorfer, M. "Ketones". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a15_077. ISBN 978-3527306732.
- ↑ Claisen, Ludwig (1896). "Ueber eine eigenthümliche Umlagerung" [On a peculiar rearrangement]. Berichte der Deutschen Chemischen Gesellschaft. 29 (3): 2931–2933. doi:10.1002/cber.189602903102.
- ↑ Spielman, M. A.; Mortenson, C. W. (1940). "The Condensation of α-Methoxystyrene with Halogen Compounds". Journal of the American Chemical Society. 62 (6): 1609–1610. doi:10.1021/ja01863a076.
- ↑ "propiophenone". Merriam-Webster.com. Merriam-Webster. Retrieved 2 June 2012.
- ↑ Hartung, Walter H.; Crossley, Frank (1936). "Isonitrosopropiophenone". Organic Syntheses. 16: 44. doi:10.15227/orgsyn.016.0044.