 | |
 | |
| Names | |
|---|---|
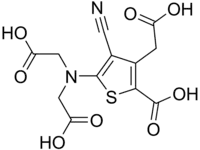
| IUPAC name
3-(Carboxymethyl)-5-[N-(carboxymethyl)glycino]-4-cyanothiophene-2-carboxylic acid | |
| Systematic IUPAC name
5-[Bis(carboxymethyl)amino]-3-(carboxymethyl)-4-cyanothiophene-2-carboxylic acid[1] | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H10N2O8S | |
| Molar mass | 342.28 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Ranelic acid is an organic acid capable of chelating metal cations.[2]
It forms the ranelate ion, C12H6N2O8S4−. Strontium ranelate, the strontium salt of ranelic acid, is a drug used to treat osteoporosis and increase bone mineral density (BMD).
References
- ↑ CID 3052774 from PubChem
- ↑ EP 415850, Wierzbicki, Michel; Bonnet, Jacqueline & Brisset, Martine et al., "Bivalent metal salts of 2-N,N-di(carboxymethyl)amino,3-cyano,4-carboxymethyl,5-carboxy-thiophene-acid, process for their preparation and pharmaceutical compositions containing them", published 1991-03-06, assigned to Adir & Co.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.