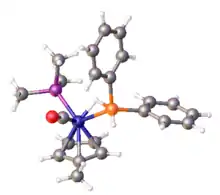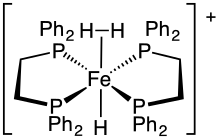
This dihydrogen complex ([HFe(H2)(dppe)2]+) is an example of a sigma complex.
In chemistry, a sigma complex or σ-complex usually refers to a family of coordination complexes where one or more ligand interacts with the metal using the bonding electrons in a sigma bond. Dihydrogen complexes are examples. Transition metal silane complexes are often especially stable sigma complexes. A subset of sigma complexes are agostic complexes, where a C-H sigma bond functions as the donor ligand. In some cases, even C-C bonds function as sigma ligands. Sigma complexes are of great mechanistic significance, despite their frequent fragility. They represent an initial interaction between the metal center and saturated substrates. Sigma complexes are generally assumed to be intermediates prior to full oxidative addition.[1]
References
- ↑ Kubas, Gregory J. (2001-08-31). Metal Dihydrogen and σ-Bond Complexes: Structure, Theory, and Reactivity. Kluwer. ISBN 0-306-46465-9.
- ↑ Schubert, U.; Scholz, G.; Müller, J.; Ackermann, K.; Wörle, B.; Stansfield, R.F.D. (1986). "Hydrido-silyl-Komplexe". Journal of Organometallic Chemistry. 306 (3): 303–326. doi:10.1016/S0022-328X(00)98993-9.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.