 | |
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
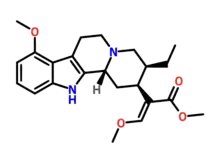
| Formula | C23H30N2O4 |
| Molar mass | 398.503 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 104 °C[1] |
| |
| |
Speciociliatine is a major alkaloid of the plant Mitragyna speciosa, commonly known as kratom. It is a stereoisomer of Mitragynine and constitutes 0.00156 - 2.9% of the dried leaf material.[2][3]
Pharmacology
Pharmacodynamics
Speciociliatine has found to be a ligand of the mu and kappa opioid receptors, however findings are varied as to whether it functions as an agonist or a competitive antagonist at those sites.[4][5]
Pharmacokinetics
A preliminary pharmacokinetic analysis in male Sprague Dawley rats determined the elimination half-life of Speciociliatine to be 2.6 - 5 hours and the absolute bioavailability to be 20.7% (at an oral dose of 20 mg/kg).[6]
References
- ↑ "National Center for Biotechnology Information (2022). PubChem Compound Summary for CID 15560576, Speciociliatine".
- ↑ Manwill, P. K., Flores-Bocanegra, L., Khin, M., Raja, H. A., Cech, N. B., Oberlies, N. H., Todd, D. A. (2022), "Kratom (Mitragyna speciosa) Validation: Quantitative Analysis of Indole and Oxindole Alkaloids Reveals Chemotypes of Plants and Products", Planta Medica, Georg Thieme Verlag KG, 88 (9/10): 838–857, doi:10.1055/a-1795-5876, PMC 9343938, PMID 35468648
- ↑ Sharma, A., Kamble, S. H., León, F., Chear, N. J. ‐Y., King, T. I., Berthold, E. C., Ramanathan, S., McCurdy, C. R., Avery, B. A. (2019), "Simultaneous quantification of ten key Kratom alkaloids in Mitragyna speciosa leaf extracts and commercial products by ultra‐performance liquid chromatography−tandem mass spectrometry", Drug Testing and Analysis, Wiley, 11 (8): 1162–1171, doi:10.1002/dta.2604, PMC 7927418, PMID 30997725
- ↑ Obeng, S., Kamble, S. H., Reeves, M. E., Restrepo, L. F., Patel, A., Behnke, M., Chear, N. J.-Y., Ramanathan, S., Sharma, A., León, F., Hiranita, T., Avery, B. A., McMahon, L. R., McCurdy, C. R. (2019), "Investigation of the Adrenergic and Opioid Binding Affinities, Metabolic Stability, Plasma Protein Binding Properties, and Functional Effects of Selected Indole-Based Kratom Alkaloids", Journal of Medicinal Chemistry, American Chemical Society (ACS), 63 (1): 433–439, doi:10.1021/acs.jmedchem.9b01465, PMC 7676998, PMID 31834797
- ↑ Kruegel, A. C., Gassaway, M. M., Kapoor, A., Váradi, A., Majumdar, S., Filizola, M., Javitch, J. A., Sames, D. (2016), "Synthetic and Receptor Signaling Explorations of the Mitragyna Alkaloids: Mitragynine as an Atypical Molecular Framework for Opioid Receptor Modulators", Journal of the American Chemical Society, American Chemical Society (ACS), 138 (21): 6754–6764, doi:10.1021/jacs.6b00360, PMC 5189718, PMID 27192616
- ↑ Berthold, E. C., Kamble, S. H., Raju, K. S., King, T. I., Popa, R., Sharma, A., León, F., Avery, B. A., McMahon, L. R., McCurdy, C. R. (2021), "Preclinical pharmacokinetic study of speciociliatine, a kratom alkaloid, in rats using an UPLC-MS/MS method", Journal of Pharmaceutical and Biomedical Analysis, Elsevier BV, 194: 113778, doi:10.1016/j.jpba.2020.113778, PMID 33277117, S2CID 227296714
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.