 | |
| Names | |
|---|---|
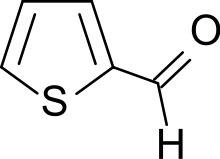
| Preferred IUPAC name
Thiophene-2-carbaldehyde | |
| Other names
2-formylthiophene, thiophene-2-aldehyde, T2A, 2-thiophenecarboxaldehyde | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.391 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C5H4OS | |
| Molar mass | 112.15 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.2 g/mL |
| Boiling point | 198 °C (388 °F; 471 K) |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H302, H315, H317, H319, H335 | |
| P261, P264, P270, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P333+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Thiophene-2-carboxaldehyde is an organosulfur compound with the formula C4H3SCHO. It is one of two isomeric thiophenecarboxaldehydes. It is a colorless liquid that often appears amber after storage. It is versatile precursor to many drugs including eprosartan, Azosemide, and Teniposide.
Preparation
It can be prepared from thiophene by the Vilsmeier reaction.[1] Alternatively, it is prepared from chloromethylation of thiophene.[2]
References
- ↑ Jonathan Swanston (2006). "Thiophene". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a26_793.pub2. ISBN 978-3527306732.
- ↑ Kenneth B. Wiberg. "2-Thiophenealdehyde". Org. Synth. 3: 811. doi:10.15227/orgsyn.000.0005.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.