Topical fluorides are fluoride-containing drugs indicated in prevention and treatment of dental caries, particularly in children's primary dentitions.[1] The dental-protecting property of topical fluoride can be attributed to multiple mechanisms of action, including the promotion of remineralization of decalcified enamel, the inhibition of the cariogenic microbial metabolism in dental plaque and the increase of tooth resistance to acid dissolution.[2] Topical fluoride is available in a variety of dose forms, for example, toothpaste, mouth rinses, varnish and silver diamine solution.[3] These dosage forms possess different absorption mechanisms and consist of different active ingredients.[4] Common active ingredients include sodium fluoride, stannous fluoride, silver diamine fluoride.[4] These ingredients account for different pharmacokinetic profiles, thereby having varied dosing regimes and therapeutic effects.[4] A minority of individuals may experience certain adverse effects, including dermatological irritation, hypersensitivity reactions, neurotoxicity and dental fluorosis.[5][6][7] In severe cases, fluoride overdose may lead to acute toxicity.[6] While topical fluoride is effective in preventing dental caries, it should be used with caution in specific situations to avoid undesired side effects.[8]
Medical uses
Topical fluoride formulations are effective measures for preventing and arresting the progression of dental caries, especially early childhood caries (ECC).[9] Domestic products such as toothpaste and mouthwash can be used on a regular basis at home, while silver diamine solution therapy can be administered by specialists in dental clinics.[9]
Mechanism of action
Topical fluoride serves to prevent early dental caries primarily in three ways: promoting remineralization of decalcified enamel, inhibiting the cariogenic microbial processes in dental plaque and increasing tooth resistance to acid breakdown.[2]
Promotion of remineralization of decalcified enamel
Fluoride has a high tendency to react with the calcium hydroxyapatite Ca10(PO4)6(OH)2 in tooth enamel due to its high affinity to metals.[8][10] It subsequently replaces the hydroxide group in hydroxyapatite to precipitate calcium fluorapatite Ca5(PO4)3)F.[8] These fluorapatite precipitations scavenge excess phosphate and calcium in the saliva to form a supersaturated solution for remineralization.[10]
Inhibition of the cariogenic microbial processes in dental plaque
Topical fluoride also serves as an antimicrobial agent to reduce demineralization by inhibiting the growth of tooth-erupting microorganisms in dental plaque.[2] Fluoride ions readily combine with hydrogen cations to produce hydrogen fluoride.[2] Hydrogen fluoride subsequently acidifies the bacterial cytoplasm, inactivating the essential enzymes for bacterial metabolism, including enolase and proton releasing adenosine triphosphatase.[2]
As topical fluoride lowers the pH, bacteria have to consume more energy to maintain a neutral environment, leaving less energy for reproduction, and further generation of polysaccharides and acids.[2] These polysaccharides are necessary for adherence to enamel, while these acids are essential for the synthesis of bacterial enzymes, for example, immunoglobulin A protease.[2] These processes contribute to reducing the risk of dental caries by inhibiting microbial metabolism in the tooth plaque.[2]
Increase in tooth resistance to acid dissolution
Topical fluoride can increase the resistance of enamel to acid.[8] Bacteria in enamel, including Streptococcus mutans, generate acids to maintain a low pH environment during fermentation.[8] These acids eventually dissociate the hydroxyapatite in teeth once the acidity falls below the critical pH (pH 5.5).[8] The fluorapatite formed by topical fluorides has lower critical pH (pH 4.5) than normal enamel, it is therefore more acid resistant and not prone to degrade even in an acidic environment.[8] This mechanism helps decelerate the rate of teeth demineralization.[8]
Dosage forms
Toothpaste

The daily use of fluoride-containing toothpaste is recognized as the key factor contributing to the global reduction in dental caries over recent decades.[11] Fluoride-containing toothpaste can be classified into two types, namely low-fluoride and high-fluoride toothpaste.[12] Low-fluoride toothpaste, depending on brand, generally contains 0.22% to 0.31% fluoride.[12] These fluorides are often manufactured in the form of sodium fluoride, stannous fluoride, or sodium monofluorophosphate (MFP).[4] High-fluoride toothpaste typically contains 1.1% sodium fluoride, namely four times more concentrated than low-fluoride toothpaste.[12] People using high-fluoride toothpaste should avoid eating or rinsing their mouth for at least 30 minutes after treatment for maximal therapeutic effect. Some fluoride-containing toothpaste incorporates extra chemical ingredients for additional purposes.[13] For instance, calcium carbonate and magnesium carbonate are added as abrasives to remove dental plaque on teeth, while strontium chloride and potassium nitrate are added as anti-sensitive agents for individuals who have teeth sensitivity.[13][14]
Mouth rinse
Fluoride mouth rinse is usually used for adjunctive therapy with other topical fluoride products.[15] It is generally prepared in the form of sodium chloride.[12] Sodium chloride is kept in the saliva after spitting out the mouth rinse, thus helping to prevent tooth decay.[12] 0.02% fluoride mouth rinse rinse is commonly administered twice daily, while 0.05% is administered once daily at bedtime after thoroughly brushing teeth.[16] People using high-fluoride toothpaste should avoid eating or rinsing their mouth for at least 30 minutes after administration for maximal therapeutic effect.[12]
Silver diamine solution

Silver diamine fluoride (SDF) is a transparent solution prepared by dissolving silver ions and fluoride ions in ammonia water.[9][1] It is approved in a few places, including Hong Kong, China, and the United States, for the prevention of early childhood caries (ECC) and relieving tooth sensitivity.[9][17]
SDF has multiple advantages over traditional fluoride varnish therapy:
- SDF is a non-invasive treatment with higher acceptability among children and elderlies.[17]
- The materials required for SDF are inexpensive, reducing the financial burden on patients.[18]
- There is currently no evidence that SDF causes serious adverse reactions, for example, acute toxicity and infection of the dental pulp, rendering it a safer therapy.[9]
- SDF followed by stannous fluoride was proven to be more effective in reducing dental caries in children's primary molars.
However, the SDF solution results in permanent black staining on the teeth's decayed proportion. This may be unacceptable by some individuals with aesthetic concerns.[9]
SDF, in addition to performing the functions of conventional topical fluorides, is suggested to have collagen-conserving properties and an additional antibacterial action owing to the presence of silver.[1] While multiple clinical trials demonstrate that 38% SDF is more effective than 5% sodium fluoride varnish in preventing ECC, it is currently unavailable in many countries due to insufficient research data.[9][17]
Adverse effects
Increased exposure of fluoride may lead to certain adverse side effects, including dental fluorosis and developmental neurotoxicity.[5][6] Other rare side effects include skin rash and hypersensitivity reactions.[7] In severe cases, fluoride overdose may lead to acute toxicity.[6][19]

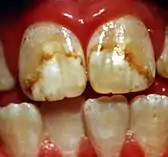
Dental fluorosis
Dental fluorosis is a dose-dependent adverse drug effect featured by temporary white marks.[5] It can be induced by increased fluoride exposure, typically from stannous fluoride-containing products or fluoridated water.[5][20] Excess intake of fluoride leads to overabundance of structurally-weak fluorapatite formed inside the enamel, resulting in increased brittleness of teeth.[21] In severe dental fluorosis, brown or yellow staining may appear on teeth.[8] Children under the age of eight are susceptible to dental fluorosis.[8]
Developmental neurotoxicity
Overdose of fluoride can potentially cause neurotoxicity during early development.[6] While the exact pathophysiology of fluoride-induced developmental toxicity is not completely understood, most research suggested that excessive fluoride intake may result in formation of aluminium fluoride (AlF3 or AlF4).[8][6] Aluminium fluoride structurally mimics phosphate, thus is capable of crossing the blood-brain barrier via phosphate transporters.[6] These fluorides in the brain may cause neurodegenerative disorders, including Alzheimer's disease, Parkinson's disease and IQ declination.[8] Nevertheless, topical fluoride was less likely to cause developmental neurotoxicity than fluoridated water.[6]
Acute fluoride toxicity
Fluoride overdose may cause acute toxicity.[6] While the underlying mechanism of fluoride toxicity is unclear, most studies ascribe fluoride toxicity to its capacity to inhibit metalloproteins by imitating metallofluoride substrate.[6] Inhibition of metalloproteins slows down multiple signalling pathways and disrupts cellular organelles, subsequently producing oxidative stress and cell cycle arrest.[6] Fluoride overload is suggested to be linked to pH and electrolyte imbalances, creating an environment unfavourable for cell living.[6] These mechanisms can ultimately result in cellular malfunction and cell death.[6]
Cautions
Toothpaste, cream, mouthrinse and varnish
- Most topical fluoride preparations with a concentration exceeding 0.6 ppm should be avoided to reduce risk of dental fluorosis if drinking water has already been fluoridated.[8][2]
- Swallowing of topical fluoride products should be avoided in order to avoid systemic adverse effects, for example, skeletal fluorosis.[22]
- While an appropriate amount of fluoride consumption during pregnancy is beneficial to prevent early childhood caries (ECC), pregnant women should avoid excessive fluoride exposure since it may predispose their children to skeletal fluorosis in later childhood.[23][24]
- Most topical fluoride preparations containing more than 1.1ppm should be avoided in children younger than 6 years of age, unless otherwise instructed by a healthcare practitioner.[25]
- Topical fluoride preparation containing benzyl alcohol derivative, polysorbate 80 and propylene glycol should be used in caution. These ingredients may precipitate severe adverse effects in neonates.[26][27][28][29]
| Ingredients | Severe adverse effects |
| Benzyl alcohol derivative |
|
| Polysorbate 80 |
|
| Propylene glycol |
|
Silver diamine fluoride
- Silver diamine fluoride is contraindicated in patients having silver allergy, oral ulcerations and severe gum disease.[9] These diseases can cause painful responses when associated with the acid or ammonia in SDF.[9]
References
- 1 2 3 Crystal, Yasmi O.; Niederman, Richard (2019-01-01). "Evidence-Based Dentistry Update on Silver Diamine Fluoride". Dental Clinics of North America. 63 (1): 45–68. doi:10.1016/j.cden.2018.08.011. PMC 6500430. PMID 30447792.
- 1 2 3 4 5 6 7 8 9 Aoun, Antoine; Darwiche, Farah; Hayek, Sibelle Al; Doumit, Jacqueline (2018-09-30). "The Fluoride Debate: The Pros and Cons of Fluoridation". Preventive Nutrition and Food Science. 23 (3): 171–180. doi:10.3746/pnf.2018.23.3.171. ISSN 2287-1098. PMC 6195894. PMID 30386744.
- ↑ Chen, Kitty; Gao, Sherry; Duangthip, Duangporn; Lo, Edward; Chu, Chun (2018-01-30). "Managing Early Childhood Caries for Young Children in China". Healthcare. 6 (1): 11. doi:10.3390/healthcare6010011. ISSN 2227-9032. PMC 5872218. PMID 29385684.
- 1 2 3 4 Nóbrega, Diego Figueiredo; Fernández, Constanza Estefany; Del Bel Cury, Altair Antoninha; Tenuta, Livia Maria Andaló; Cury, Jaime Aparecido (2016). "Frequency of Fluoride Dentifrice Use and Caries Lesions Inhibition and Repair". Caries Research. 50 (2): 133–140. doi:10.1159/000444223. ISSN 0008-6568. PMID 26992247. S2CID 11785219.
- 1 2 3 4 Di Giovanni, Tamara; Eliades, Theodore; Papageorgiou, Spyridon N. (2018-11-01). "Interventions for dental fluorosis: A systematic review". Journal of Esthetic and Restorative Dentistry. 30 (6): 502–508. doi:10.1111/jerd.12408. PMID 30194793. S2CID 52172575.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 Grandjean, Philippe (2019-12-01). "Developmental fluoride neurotoxicity: an updated review". Environmental Health. 18 (1): 110. doi:10.1186/s12940-019-0551-x. ISSN 1476-069X. PMC 6923889. PMID 31856837.
- 1 2 Cvikl, Barbara; Moritz, Andreas; Bekes, Katrin (2018-06-12). "Pit and Fissure Sealants—A Comprehensive Review". Dentistry Journal. 6 (2): 18. doi:10.3390/dj6020018. ISSN 2304-6767. PMC 6023524. PMID 29895726.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 Johnston, Nichole R.; Strobel, Scott A. (2020-04-01). "Principles of fluoride toxicity and the cellular response: a review". Archives of Toxicology. 94 (4): 1051–1069. doi:10.1007/s00204-020-02687-5. ISSN 0340-5761. PMC 7230026. PMID 32152649.
- 1 2 3 4 5 6 7 8 9 Duangthip, Duangporn; Chen, Kitty; Gao, Sherry; Lo, Edward; Chu, Chun (2017-10-10). "Managing Early Childhood Caries with Atraumatic Restorative Treatment and Topical Silver and Fluoride Agents". International Journal of Environmental Research and Public Health. 14 (10): 1204. doi:10.3390/ijerph14101204. ISSN 1660-4601. PMC 5664705. PMID 28994739.
- 1 2 Farooq, Imran; Bugshan, Amr (2021-06-15). "The role of salivary contents and modern technologies in the remineralization of dental enamel: a narrative review". F1000Research. 9: 171. doi:10.12688/f1000research.22499.3. ISSN 2046-1402. PMC 7076334. PMID 32201577.
- ↑ Pitts, Nigel B.; Zero, Domenick T.; Marsh, Phil D.; Ekstrand, Kim; Weintraub, Jane A.; Ramos-Gomez, Francisco; Tagami, Junji; Twetman, Svante; Tsakos, Georgios; Ismail, Amid (2017-12-21). "Dental caries". Nature Reviews Disease Primers. 3 (1): 17030. doi:10.1038/nrdp.2017.30. ISSN 2056-676X. PMID 28540937.
- 1 2 3 4 5 6 Hua, Yong-Mei; Chen, Jie; Jean, Gong (2006-04-01). "[The preventive effectiveness in reducing tooth decay and decalcification of different concentration of fluoride toothpaste for orthodontic patients]". Hua Xi Kou Qiang Yi Xue Za Zhi = Huaxi Kouqiang Yixue Zazhi = West China Journal of Stomatology. 24 (2): 146–147. ISSN 1000-1182. PMID 16704094.
- 1 2 Epple, Matthias; Meyer, Frederic; Enax, Joachim (2019-08-01). "A Critical Review of Modern Concepts for Teeth Whitening". Dentistry Journal. 7 (3): 79. doi:10.3390/dj7030079. ISSN 2304-6767. PMC 6784469. PMID 31374877.
- ↑ Chen, Lijie; Al-Bayatee, Suma; Khurshid, Zohaib; Shavandi, Amin; Brunton, Paul; Ratnayake, Jithendra (2021-08-27). "Hydroxyapatite in Oral Care Products—A Review". Materials. 14 (17): 4865. Bibcode:2021Mate...14.4865C. doi:10.3390/ma14174865. ISSN 1996-1944. PMC 8432723. PMID 34500955.
- ↑ Keller, M. K.; Klausen, B. J.; Twetman, S. (2016-03-01). "Fluoride varnish or fluoride mouth rinse? A comparative study of two school-based programs". Community Dental Health. 33 (1): 23–26. ISSN 0265-539X. PMID 27149769.
- ↑ Azcurra, A. I.; Calamari, S. E.; Yankilevich, E. R.; Battellino, L. J.; Cattoni, S. T.; Colantonio, G. (1997). "[Effects of local treatment with sodium fluoride mouthrinse on peroxidase and hypothiocyanite saliva levels in adolescent]/". Acta Physiologica, Pharmacologica et Therapeutica Latinoamericana. 47 (4): 211–220. ISSN 0327-6309. PMID 9504181.
- 1 2 3 Horst, J.A. (2018-02-01). "Silver Fluoride as a Treatment for Dental Caries". Advances in Dental Research. 29 (1): 135–140. doi:10.1177/0022034517743750. ISSN 0895-9374. PMC 6699125. PMID 29355428.
- ↑ Oliveira, Branca Heloisa; Cunha-Cruz, Joana; Rajendra, Anjana; Niederman, Richard (2018-08-01). "Controlling caries in exposed root surfaces with silver diamine fluoride". The Journal of the American Dental Association. 149 (8): 671–679.e1. doi:10.1016/j.adaj.2018.03.028. PMC 6064675. PMID 29805039.
- ↑ Qiao, Lichun; Liu, Xuan; He, Yujie; Zhang, Jiaheng; Huang, Hao; Bian, Wenming; Chilufya, Mumba Mulutula; Zhao, Yan; Han, Jing (2021-11-03). "Progress of Signaling Pathways, Stress Pathways and Epigenetics in the Pathogenesis of Skeletal Fluorosis". International Journal of Molecular Sciences. 22 (21): 11932. doi:10.3390/ijms222111932. ISSN 1422-0067. PMC 8584317. PMID 34769367.
- ↑ Wang, Feiqing; Li, Yanju; Tang, Dongxin; Zhao, Jianing; Yang, Xu; Liu, Yanqing; Peng, Fengtao; Shu, Liping; Wang, Jishi; He, Zhixu; Liu, Yang (2021-02-01). "Effects of water improvement and defluoridation on fluorosis-endemic areas in China: A meta-analysis". Environmental Pollution. 270: 116227. doi:10.1016/j.envpol.2020.116227. PMID 33333408. S2CID 229319170.
- ↑ Wei, Wei; Pang, Shujuan; Sun, Dianjun (2019-04-01). "The pathogenesis of endemic fluorosis: Research progress in the last 5 years". Journal of Cellular and Molecular Medicine. 23 (4): 2333–2342. doi:10.1111/jcmm.14185. ISSN 1582-1838. PMC 6433665. PMID 30784186.
- ↑ Mascarenhas, Ana Karina; Burt, Brian A. (1998-08-01). "Fluorosis risk from early exposure to fluoride toothpaste". Community Dentistry and Oral Epidemiology. 26 (4): 241–248. doi:10.1111/j.1600-0528.1998.tb01957.x. hdl:2027.42/75437. ISSN 0301-5661. PMID 9758424.
- ↑ Yildiz, Mustafa; Oral, Baha (2006-06-01). "The effect of pregnancy and lactation on bone mineral density in fluoride-exposed rats". Toxicology and Industrial Health. 22 (5): 217–222. doi:10.1191/0748233706th258oa. ISSN 0748-2337. PMID 16898264. S2CID 27189533.
- ↑ Wegehaupt, Florian; Menghini, Giorgio (2020-09-07). "Fluoride Update". Swiss Dental Journal. 130 (9): 677–683. ISSN 2296-6498. PMID 32893610.
- ↑ Shulman, Jay D.; Wells, Linda M. (1997-09-01). "Acute Fluoride Toxicity from Ingesting Home-use Dental Products in Children, Birth to 6 Years of Age". Journal of Public Health Dentistry. 57 (3): 150–158. doi:10.1111/j.1752-7325.1997.tb02966.x. ISSN 0022-4006. PMID 9383753.
- ↑ Gershanik, Juan; Boecler, Betty; Ensley, Harry; McCloskey, Sharon; George, William (1982-11-25). "The Gasping Syndrome and Benzyl Alcohol Poisoning". New England Journal of Medicine. 307 (22): 1384–1388. doi:10.1056/NEJM198211253072206. ISSN 0028-4793. PMID 7133084.
- ↑ Gernhardt, Christian Ralf; Bekes, Katrin; Schaller, Hans-Guenter (2007-12-01). "Influence of three different sealants on root dentin demineralization in situ". American Journal of Dentistry. 20 (6): 390–393. ISSN 0894-8275. PMID 18269131.
- ↑ Gerasimidis, Konstantinos; Bryden, Katie; Chen, Xiufen; Papachristou, Eleftheria; Verney, Anais; Roig, Marine; Hansen, Richard; Nichols, Ben; Papadopoulou, Rodanthi; Parrett, Alison (2020-10-01). "The impact of food additives, artificial sweeteners and domestic hygiene products on the human gut microbiome and its fibre fermentation capacity". European Journal of Nutrition. 59 (7): 3213–3230. doi:10.1007/s00394-019-02161-8. ISSN 1436-6207. PMC 7501109. PMID 31853641.
- ↑ Skaare, Anne; Kjærheim, Vibeke; Barkvoll, Pål; Rølla, Gunnar (1997-01-01). "Skin Reactions and Irritation Potential of Four Commercial Toothpastes". Acta Odontologica Scandinavica. 55 (2): 133–136. doi:10.3109/00016359709115405. ISSN 0001-6357. PMID 9176662.