 | |
| Clinical data | |
|---|---|
| Other names | URP |
| Physiological data | |
| Source tissues | organum vasculosum laminae terminalis; median eminence |
| Target tissues | system-wide |
| Receptors | Urotensin II Receptor |
| Agonists | Direct: Urotensin-II |
| Antagonists | BIM23127, GSK 1562590 hydrochloride, SB 611812, SB 657510, SB 706375, UFP 803, Urantide, Orn5-URP |
| Precursor | Prepro-URP |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| KEGG | |
Urotensin II-related peptide (URP) is a cyclic neuropeptide that is found in all vertebrates that have been genome sequenced so far. It has a long lasting hypotensive effect and may also regulate reproduction. It is part of the Urotensin II system and is one of the two endogenous ligands for rats, mice, and possibly humans.
Discovery
URP was discovered in rats when researchers were trying to locate urotensin II (UII), a neuropeptide that is a potent vasoconstrictor and increases REM cycles in the brain. The researchers designed antibodies using Goby UII as an antigen that would target the specific peptide sequence CFWKYC. When the peptide was observed using a mass spectrometer the scientists discovered this peptide was smaller than UII but had similar characteristics as well which is why it was called Urotensin II-related peptide.[1]
Structure
The URP gene is located on the 3q28 chromosome of humans. The mature URP peptide is only 8 peptides long making it smaller than UII. URP is also the same across all vertebrates because it has the same cleaving site unlike UII whose cleaving sites vary among species making its sequence different for all species.[1] URP has the same cysteine bridged hexapeptide ring with the sequence CFWKYC as UII. This is known as the core and is the major site of action on the peptide. Destruction of the core leads to immediate loss of biological activity.[2] On the other hand, the amino terminus of URP doesn’t seem to contain any relevant information because it can be modified without any loss in pharmacological activity.[3] Unlike UII, URP doesn’t have an acidic amino acid (either glutamic acid or aspartic acid) preceding its core. It is still a potent agonist for the UII- receptor which suggests that this acidic amino acid is not required for activation of the receptor.
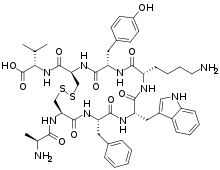
The peptide sequence for URP is:
Receptor
URP is an agonist for the UII receptor which is a G protein-coupled receptor with the alpha subunit Gαq11. This activates PKC which then activates PLC which increases the intercellular calcium concentration. It is found in many peripheral tissues, blood vessels, and also the brainstem cholinergic neurons of the laterodorsal tegmental (LDT) and the pedunculopontine tegmental nuclei (PPT).[4]
Tissue Localization
Prepro-URP which is the precursor to the mature URP peptide is found in various tissues including specific parts of the brain such as frontal lobe and hypothalamus, and other peripheral tissues such as heart, kidneys, lungs, placenta, ovaries, and testes. In humans the amount of UII and URP gene expression are comparable except in the spinal cord where UII gene expression is much higher.[1]
In rats the UII gene expression is higher than the URP gene expression throughout the entire body. However, when the brains of the rats were tested, only the URP peptide was found making it the primary endogenous ligand in the brain.[1]
Unlike humans and rats, URP gene expression is found in mice spinal cords.[5]
Function
Cardiovascular
When URP is injected into rats a long hypotensive response will be observed.[1] UII is known as a vasoconstrictor meaning that even though both are agonists for the same receptor they can produce opposite effects[6]
CNS
Axons that react to URP are primarily found in organum vasculosum laminae terminalis (OVLT) and in the median eminence (ME). These axons are located near the hypothalamus and almost always contain the hormone Gondotropin- releasing hormone (GnRH) which was found through in situ hybridization which provides information of the anatomical location URP mRNA. This means that URP might have an effect on reproduction which has not been discovered .[5]
Binding between UII and URP
Since they are both ligands for the same receptor, an experiment was done to determine which ligand had a higher affinity. When the binding of the two were compared and tested, URP actually had higher affinity.[1]
References
- 1 2 3 4 5 6 7 Sugo T, Murakami Y, Shimomura Y, Harada M, Abe M, Ishibashi Y, et al. (October 2003). "Identification of urotensin II-related peptide as the urotensin II-immunoreactive molecule in the rat brain". Biochemical and Biophysical Research Communications. 310 (3): 860–868. doi:10.1016/j.bbrc.2003.09.102. PMID 14550283.
- ↑ McMaster D, Kobayashi Y, Rivier J, Lederis K (1986). "Characterization of the biologically and antigenically important regions of urotensin II". Proceedings of the Western Pharmacology Society. 29: 205–208. PMID 3763612.
- ↑ Coy DH, Rossowski WJ, Cheng BL, Taylor JE (December 2002). "Structural requirements at the N-terminus of urotensin II octapeptides". Peptides. 23 (12): 2259–2264. doi:10.1016/s0196-9781(02)00266-8. PMID 12535707. S2CID 23960477.
- ↑ Clark SD, Nothacker HP, Wang Z, Saito Y, Leslie FM, Civelli O (December 2001). "The urotensin II receptor is expressed in the cholinergic mesopontine tegmentum of the rat". Brain Research. 923 (1–2): 120–127. doi:10.1016/s0006-8993(01)03208-5. PMID 11743979. S2CID 29248060.
- 1 2 Egginger JG, Calas A (August 2005). "A novel hypothalamic neuroendocrine peptide: URP (urotensin-II-related peptide)?". Comptes Rendus Biologies. 328 (8): 724–731. doi:10.1016/j.crvi.2005.06.002. PMID 16125650.
- ↑ Saetrum Opgaard O, Nothacker H, Ehlert FJ, Krause DN (October 2000). "Human urotensin II mediates vasoconstriction via an increase in inositol phosphates". European Journal of Pharmacology. 406 (2): 265–271. doi:10.1016/s0014-2999(00)00672-5. PMID 11020490.
Further reading
- Sugo T, Mori M (May 2008). "Another ligand fishing for G protein-coupled receptor 14. Discovery of urotensin II-related peptide in the rat brain". Peptides. 29 (5): 809–812. doi:10.1016/j.peptides.2007.06.005. PMID 17628210. S2CID 13387250.
- Nothacker HP, Clark S (November 2005). "From heart to mind. The urotensin II system and its evolving neurophysiological role". The FEBS Journal. 272 (22): 5694–5702. doi:10.1111/j.1742-4658.2005.04983.x. PMID 16279935.