 | |
| Names | |
|---|---|
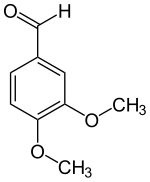
| Preferred IUPAC name
3,4-Dimethoxybenzaldehyde | |
| Systematic IUPAC name
3,4-Dimethoxybenzenecarbaldehyde | |
| Other names
Methylvanillin; Veratric aldehyde; Veratral; Veratryl aldehyde; Veratrum aldehyde; Vanillin methyl ether | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.976 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H10O3 | |
| Molar mass | 166.176 g·mol−1 |
| Appearance | Peach coloured crystals |
| Density | 1.114 g/mL |
| Melting point | 40 to 43 °C (104 to 109 °F; 313 to 316 K) |
| Boiling point | 281 °C (538 °F; 554 K) |
| organic solvents | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Harmful |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Veratraldehyde (3,4-dimethoxybenzaldehyde) is an organic compound that is widely used as a flavorant and odorant. The compound is structurally related to benzaldehyde.
This compound is popular commercially because of its pleasant woody fragrance. It is derivative of vanillin, from which it is prepared by methylation.[1]
Uses
Veratraldehyde can be used as an intermediate in the synthesis of some pharmaceutical drugs including amiquinsin, hoquizil, piquizil, prazosin, quinazoline, tiapamil, toborinone, verazide, and vetrabutine.
See also
References
- ↑ Karl-Georg Fahlbusch, Franz-Josef Hammerschmidt, Johannes Panten, Wilhelm Pickenhagen, Dietmar Schatkowski, , Kurt Bauer, Dorothea Garbe and Horst Surburg "Flavors and Fragrances" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2003. doi:10.1002/14356007.a11_141
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.