 | |
| Names | |
|---|---|
| Preferred IUPAC name
4-Methoxy-7-methyl-5H-furo[3,2-g][1]benzopyran-5-one[1] | |
| Other names
Visnacorin; Khella; Desmethoxykhellin; 5-Methoxy-2-methylfuranochromone; 5-Methoxy-2-methyl-6,7-furanochrome | |
| Identifiers | |
3D model (JSmol) |
|
| 5-19-06-00030 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.001.301 |
| EC Number |
|
| 234955 | |
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C13H10O4 | |
| Molar mass | 230.219 g·mol−1 |
| Appearance | Solid |
| Melting point | 144 to 145 °C (291 to 293 °F; 417 to 418 K) |
| soluble | |
| Solubility | ethanol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Harmful by ingestion |
| GHS labelling: | |
 | |
| Warning | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
832 mg/kg (oral, rat) |
| Safety data sheet (SDS) | Sigma-Aldrich MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
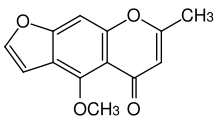
Visnagin is an organic chemical compound with the molecular formula C13H10O4 It is a furanochromone, a compound derivative of chromone (1,4-benzopyrone) and furan.
History
Visnaga daucoides, the main source for visnagin, has been used in traditional medicine in the Middle East to ease urinary tract pain associated with kidney stones and to promote stone passage.[2]
Occurrences
Visnagin naturally occurs in Visnaga daucoides, a species of flowering plant in the carrot family known by many common names, including bisnaga, toothpickweed, and khella. Visnagin-containing khella seeds are usually found mainly in Middle East countries such as Egypt and Turkey and also in Northern African countries such as Morocco. Visnagin can be extracted directly from khella seeds.
Synthesis
Modified synthesis of the naturally occurring visnagin is reported. Starting from phloroghrcin aldehyde, and building on the 2-methyl-y-pyrone, 2-methyl-5,7-dihydroxy-dfo-yl-chromone was obtained. Construction of the furan moiety was realized by a conventional method through the 7-carboxymethoxy ether giving S-norvisnagin which can be methylated to visnagin.[3]
Reactions
Condensation
Visnagin analogs can be synthesized through the condensation of visnagone with esters and sodium. This leads to the product of 2-ethyl, 2-(3'-pyridyl) visnagin analog (50c).[4]
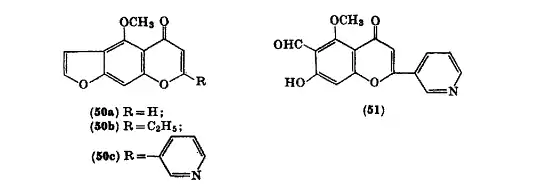 [4]
[4]
Oligomerization
Visnagin molecules can go over an oligomerization to form a chain of visnagin molecules.[5]
 Visnagin olygomerization reaction
Visnagin olygomerization reaction
Animal study
Visnagin has biological activity in animal models as a vasodilator and reduces blood pressure by inhibiting calcium influx into the cell.[6] In rats, visnagin prevents the formation of kidney stones by prolonging the induction time of nucleation of crystals.[7]
On December 8, 2014, it was reported that "visnagin protects against doxorubicin-induced cardiomyopathy through modulation of mitochondrial malate dehydrogenase."[8]
References
- ↑ Visnacorin, ChemSpider
- ↑ Haug, Karin G.; Weber, Benjamin; Hochhaus, Guenther; Butterweck, Veronika (2012). "Nonlinear pharmacokinetics of visnagin in rats after intravenous bolus administration". European Journal of Pharmaceutical Sciences. 45 (1–2): 79–89. doi:10.1016/j.ejps.2011.10.023. PMID 22085634.
Ammi visnaga L. (syn. Khella, Apiaceae) fruit preparations, such as tea prepared from crushed or powdered seeds, have traditionally been used in the Middle East to ease urinary tract pain associated with kidney stones and to promote stone passage (Gunaydin and Beyazit, 2004).
- ↑ Badawi, M.M.; Fayez, M.B.E. (1965). "Natural chromones—I". Tetrahedron. 21 (10): 2925. doi:10.1016/S0040-4020(01)98378-4.
- 1 2 Mustafa, Ahmed (2009-09-15). The Chemistry of Heterocyclic Compounds, Furopyrans and Furopyrones. John Wiley & Sons. ISBN 9780470188354.
- ↑ Pradhan, Padmanava; Banerji, Asoke (1998). "Novel cyclobutane fused furochromone oligomers from the seeds of Pimpinella monoica Dalz". Tetrahedron. 54 (48): 14541. doi:10.1016/S0040-4020(98)00913-2.
- ↑ Lee, Jin-Koo; Jung, Jun-Sub; Park, Sang-Hee; Park, Soo-Hyun; Sim, Yun-Beom; Kim, Seon-Mi; Ha, Tal-Soo; Suh, Hong-Won (2010). "Anti-inflammatory effect of visnagin in lipopolysaccharide-stimulated BV-2 microglial cells". Archives of Pharmacal Research. 33 (11): 1843–50. doi:10.1007/s12272-010-1117-1. PMID 21116788. S2CID 22168754.
- ↑ Abdel-Aal, E.A.; Daosukho, S.; El-Shall, H. (2009). "Effect of supersaturation ratio and Khella extract on nucleation and morphology of kidney stones". Journal of Crystal Growth. 311 (9): 2673. Bibcode:2009JCrGr.311.2673A. doi:10.1016/j.jcrysgro.2009.02.027.
- ↑ Liu, Y.; Asnani, A.; Zou, L.; Bentley, V. L.; Yu, M.; Wang, Y.; Dellaire, G.; Sarkar, K. S.; Dai, M.; Chen, H. H.; Sosnovik, D. E.; Shin, J. T.; Haber, D. A.; Berman, J. N.; Chao, W.; Peterson, R. T. (10 December 2014). "Visnagin protects against doxorubicin-induced cardiomyopathy through modulation of mitochondrial malate dehydrogenase". Science Translational Medicine. 6 (266): 266ra170. doi:10.1126/scitranslmed.3010189. PMC 4360984. PMID 25504881.
External links
 Media related to Visnagin at Wikimedia Commons
Media related to Visnagin at Wikimedia Commons
