JWH大麻素列表
克莱门森大学的约翰·威廉·霍夫曼(John William Huffman)研究小组合成了450多种大麻素[1][2]。其中一些是[3][4]:
| 名称 | 类别 | 分子式 | Ki/nM (CB1) | Ki/nM (CB2) | 选择性 | 结构 |
|---|---|---|---|---|---|---|
| JWH-004 | 萘甲酰基吲哚 | C26H27NO | 48 ± 13 | 4 ± 1.5 | CB2 (12x) | 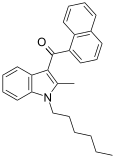 |
| JWH-007[5] | 萘甲酰基吲哚 | C25H25NO | 9.5 ± 4.5 | 2.9 ± 2.6 | CB2 (3.3x) | 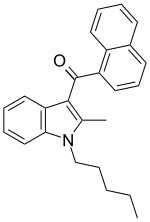 |
| JWH-009 | 萘甲酰基吲哚 | C27H29NO | >10000 | 141 ± 14 | CB2 (>70x) | 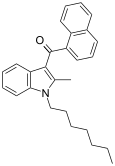 |
| JWH-011 | 萘甲酰基吲哚 | C27H29NO | 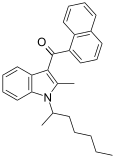 | |||
| JWH-015[5] | 萘甲酰基吲哚 | C23H21NO | 164 ± 22 | 13.8 ± 4.6 | CB2 (12x) | 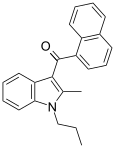 |
| JWH-016 | 萘甲酰基吲哚 | C24H23NO | 22 ± 1.5 | 4.3 ± 1.6 | CB2 (5.1x) | 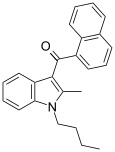 |
| JWH-018[5] | 萘甲酰基吲哚 | C24H23NO | 9 ± 5 | 2.9 ± 2.6 | CB2 (3.1x) | 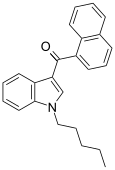 |
| JWH-019 | 萘甲酰基吲哚 | C25H25NO | 9.8 ± 2 | 5.55 ± 2 | CB2 (1.77x) | 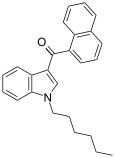 |
| JWH-020 | 萘甲酰基吲哚 | C26H27NO | 128 ± 17 | 205 ± 20 | CB1 (1.6x) | 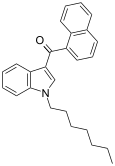 |
| JWH-030 | 萘甲酰基吡咯 | C20H21NO | 87 ± 3 | 320 ± 127 | CB1 (3.7x) | 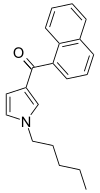 |
| JWH-031 | 萘甲酰基吡咯 | C21H23NO | 399 ± 109 | 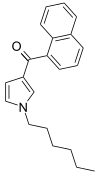 | ||
| JWH-032 | 萘甲酰基吡咯 | C18H17NO | >10000 | >10000 | — | 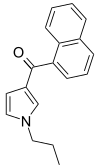 |
| JWH-033 | 萘甲酰基吡咯 | C19H19NO | 666 ± 77 | 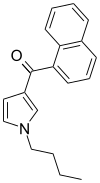 | ||
| JWH-036 | 萘甲酰基吡咯 | C22H25NO | 309 ± 11 | 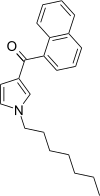 | ||
| JWH-042[6] | 萘甲酰基吲哚 | C21H17NO | >10000 | 5050 ± 192 | CB2 | 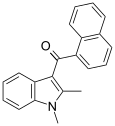 |
| JWH-043[6] | 萘甲酰基吲哚 | C22H19NO | 1180 ± 44 | 964 ± 242 | CB2 (1.2x) | 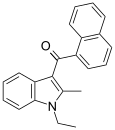 |
| JWH-044 | 萘甲酰基吡咯 | C16H13NO | >10000 | >10000 | — | 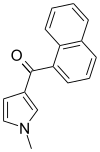 |
| JWH-045 | 萘甲酰基吡咯 | C17H15NO | >10000 | >10000 | — | 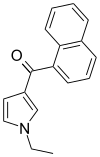 |
| JWH-046[6] | 萘甲酰基吲哚 | C24H23NO | 343 ± 38 | 16.3 ± 4.9 | CB2 (21x) | 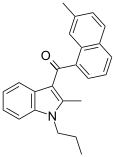 |
| JWH-047[6] | 萘甲酰基吲哚 | C25H25NO | 59 ± 3 | 3.47 ± 1.80 | CB2 (17x) | 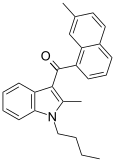 |
| JWH-048[6] | 萘甲酰基吲哚 | C26H27NO | 10.7 ± 1.0 | 0.49 ± 0.13 | CB2 (22x) | 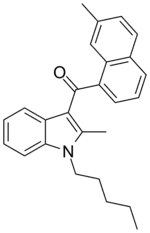 |
| JWH-049[6] | 萘甲酰基吲哚 | C27H29NO | 55.1 ± 17.0 | 32.3 ± 2.4 | CB2 (1.7x) | 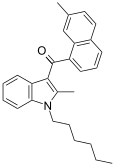 |
| JWH-050[6] | 萘甲酰基吲哚 | C28H31NO | 342 ± 6 | 526 ± 133 | CB1 (1.5x) | 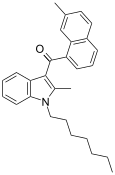 |
| JWH-051 | 二苯并吡喃 | C25H38O2 | 1.20 | 0.03 | CB2 (40x) |  |
| JWH-056[7] | 二苯并吡喃 | C21H30O | >10000 | 32 ± 9 | CB2 | 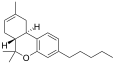 |
| JWH-057[8] | 二苯并吡喃 | C25H38O | 23 ± 7 | 2.9 ± 1.6 | CB2 (8x) | 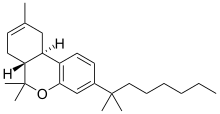 |
| JWH-065[7] | 二苯并吡喃 | C23H34O | 399 ± 76 | 10 ± 2 | CB2 (40x) | 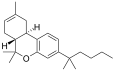 |
| JWH-070[6] | 萘甲酰基吲哚 | C20H15NO | >10000 | >10000 | 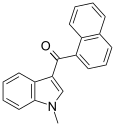 | |
| JWH-071[6] | 萘甲酰基吲哚 | C21H17NO | 1340 ± 123 | 2940 ± 852 | CB1 (2.2x) | 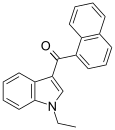 |
| JWH-072 | 萘甲酰基吲哚 | C22H19NO | 1050 ± 5.5 | 170 ± 54 | CB2 (6x) | 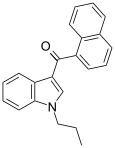 |
| JWH-073 | 萘甲酰基吲哚 | C23H21NO | 8.9 ± 1.8 | 27 ± 12 | CB1 (3x) | 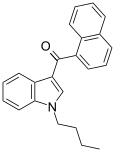 |
| JWH-076[5] | 萘甲酰基吲哚 | C23H21NO | 214 ± 11 | 106 ± 46 | CB2 (2x) | 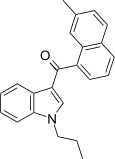 |
| JWH-077[6] | 萘甲酰基吲哚 | C21H17NO2 | >10000 | >10000 | 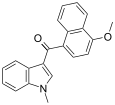 | |
| JWH-078[6] | 萘甲酰基吲哚 | C22H19NO2 | 817 ± 60 | 633 ± 116 | CB2 (1.3x) | 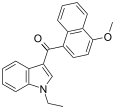 |
| JWH-079[6] | 萘甲酰基吲哚 | C23H21NO2 | 63.0 ± 3.0 | 32.0 ± 6.0 | CB2 (2x) | 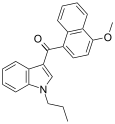 |
| JWH-080[6] | 萘甲酰基吲哚 | C24H23NO2 | 8.9 ± 1.8 | 2.21 ± 1.30 | CB2 (4x) | 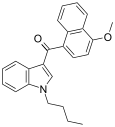 |
| JWH-081[6] | 萘甲酰基吲哚 | C25H25NO2 | 1.2 ± 0.03 | 12.4 ± 2.2 | CB1 (10x) |  |
| JWH-082[6] | 萘甲酰基吲哚 | C26H27NO2 | 5.3 ± 0.8 | 6.40 ± 0.94 | CB1 (1.2x) | 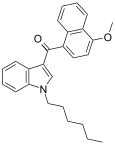 |
| JWH-083[6] | 萘甲酰基吲哚 | C27H29NO2 | 106 ± 12 | 102 ± 50 | — | 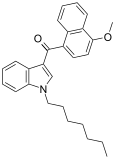 |
| JWH-091[9] (Δ8-THCP) | 二苯并吡喃 | C23H34O2 | 22.0 ± 3.9 | 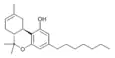 | ||
| JWH-093[6] | 萘甲酰基吲哚 | C27H29NO2 | 40.7 ± 2.8 | 59.1 ± 10.5 | CB1 (1.45x) | 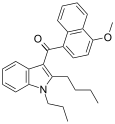 |
| JWH-094[6] | 萘甲酰基吲哚 | C24H23NO2 | 476 ± 67 | 97.3 ± 2.7 | CB2 (4.9x) | 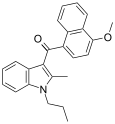 |
| JWH-095[6] | 萘甲酰基吲哚 | C29H33NO2 | 140 ± 4.3 | 312 ± 83 | CB1 (2.2x) | 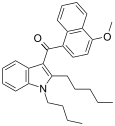 |
| JWH-096[6] | 萘甲酰基吲哚 | C25H25NO2 | 33.7 ± 2.9 | 13.3 ± 5.6 | CB2 (2.5x) | 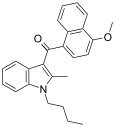 |
| JWH-097[6] | 萘甲酰基吲哚 | C31H37NO2 | 455 ± 28 | 121 ± 15 | CB2 (3.8x) | 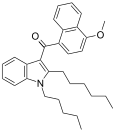 |
| JWH-098[6] | 萘甲酰基吲哚 | C26H27NO2 | 4.5 ± 0.1 | 1.9 ± 0.3 | CB2 (2.4x) | 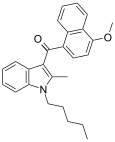 |
| JWH-099[6] | 萘甲酰基吲哚 | C27H29NO2 | 35.3 ± 9.0 | 17.8 ± 2.9 | CB2 (2x) | 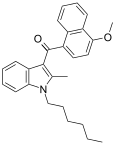 |
| JWH-100[6] | 萘甲酰基吲哚 | C25H38O2 | 381 ± 102 | 155 ± 74 | CB2 (2.5x) | 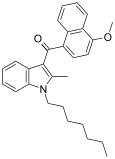 |
| JWH-102[7] | 二苯并吡喃 | C24H38O2 | 7.9 ± 0.9 | 5.2 ± 2.0 | CB2 (1.5x) | 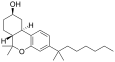 |
| JWH-103[7] | 二苯并吡喃 | C24H38O2 | 28 ± 3 | 23 ± 7 | CB2 (1.2x) | 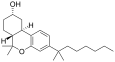 |
| JWH-116[10] | 萘甲酰基吲哚 | C26H27NO | 52 ± 5 | 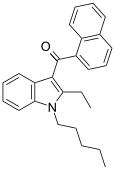 | ||
| JWH-120[5] | 萘甲酰基吲哚 | C23H21NO | 1054 ± 31 | 6.1 ± 0.7 | CB2 (173x) | 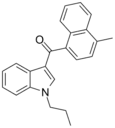 |
| JWH-121 | 萘甲酰基吲哚 | C24H23NO | — | 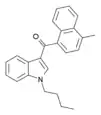 | ||
| JWH-122[10] | 萘甲酰基吲哚 | C25H25NO | 0.69 ± 0.05 | 1.2 ± 1.2 | — | 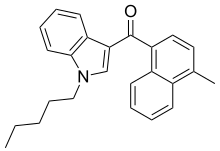 |
| JWH-124 (Δ8-Parahexyl) | 二苯并吡喃 | C22H32O2 | 41.0 ± 3.8 | 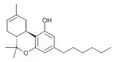 | ||
| JWH-130 (Δ8-THCB) | 二苯并吡喃 | C20H28O2 | 65.0 ± 13 | 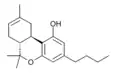 | ||
| JWH-133[7] | 二苯并吡喃 | C22H32O | 677 ± 132 | 3.4 ± 1.0 | CB2 (200x) | 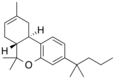 |
| JWH-138[11] | 二苯并吡喃 | C24H36O2 | 8.5 ± 1.4 | 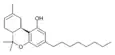 | ||
| JWH-139[12] | 二苯并吡喃 | C21H30O | 2290 ± 505 | 14 ± 10 | CB2 (164x) | 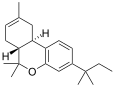 |
| JWH-142[7] | 二苯并吡喃 | C26H40O2 | 529 ± 49 | 35 ± 14 | CB2 (15x) | 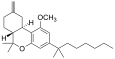 |
| JWH-143[7] | 二苯并吡喃 | C26H40O2 | 924 ± 104 | 65 ± 8 | CB2 (14x) | 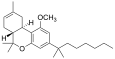 |
| JWH-145[13] | 萘甲酰基吡咯 | C26H25NO | 14 ± 2 | 6.4 ± 0.4 | CB2 (2.2x) | 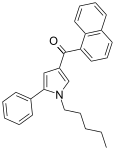 |
| JWH-146[13] | 萘甲酰基吡咯 | C28H29NO | 21 ± 2 | 62 ± 5 | CB2 (3.0x) | 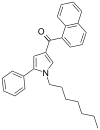 |
| JWH-147[13] | 萘甲酰基吡咯 | C27H27NO | 11 ± 1 | 7.1 ± 0.2 | CB2 (1.5x) | 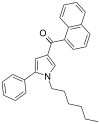 |
| JWH-148[5] | 萘甲酰基吲哚 | C24H23NO | 123 ± 8 | 14.0 ± 1.0 | CB2 (8x) | 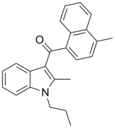 |
| JWH-149[5] | 萘甲酰基吲哚 | C26H27NO | 5.0 ± 2.1 | 0.73 ± 0.03 | CB2 (6.8x) | 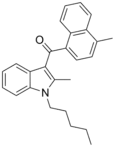 |
| JWH-150[13] | 萘甲酰基吡咯 | C25H23NO | 60 ± 1 | 15 ± 2 | CB2 (4x) | 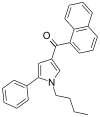 |
| JWH-151[5] | 萘甲酰基吲哚 | C24H23NO2 | >10000 | 30 ± 1.1 | CB2 (>333x) | 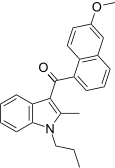 |
| JWH-153[5] | 萘甲酰基吲哚 | C26H27NO2 | 250 ± 24 | 11 ± 0.5 | CB2 (23x) | 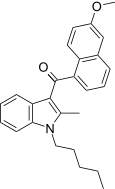 |
| JWH-156[13] | 萘甲酰基吡咯 | C24H21NO | 404 ± 18 | 104 ± 18 | CB2 (4x) | 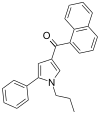 |
| JWH-159[5] | 萘甲酰基吲哚 | C26H27NO2 | 45 ± 1 | 10.4 ± 1.4 | CB2 (4.3x) | 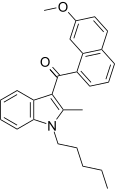 |
| JWH-160[5] | 萘甲酰基吲哚 | C24H23NO2 | 1568 ± 201 | 441 ± 110 | CB2 (3.6x) | 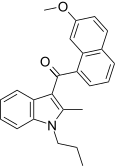 |
| JWH-161 | 二苯并吡喃合体 | 19.0 | 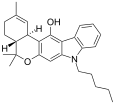 | |||
| JWH-163[5] | 萘甲酰基吲哚 | C23H21NO2 | 2358 ± 215 | 138 ± 12 | CB2 (17x) | 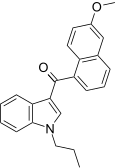 |
| JWH-164[5] | 萘甲酰基吲哚 | C25H25NO2 | 6.6 ± 0.7 | 6.9 ± 0.2 | — | 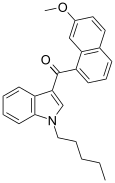 |
| JWH-165[5] | 萘甲酰基吲哚 | C23H21NO2 | 204 ± 26 | 71 ± 8 | CB2 (2.9x) | 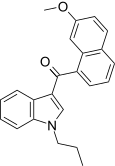 |
| JWH-166[5] | 萘甲酰基吲哚 | C25H25NO2 | 44 ± 10 | 1.9 ± 0.08 | CB2 (23x) | 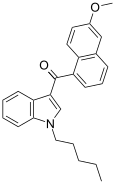 |
| JWH-167 | 苯乙酰吲哚 | C21H23NO | 90 ± 17 | 159 ± 14 | CB1 (1.77x) | 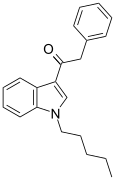 |
| JWH-171 | 碳氢 | 51 | 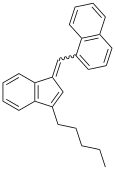 | |||
| JWH-175[10] | 萘甲基吲哚 | 22 ± 2 | 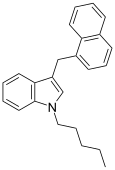 | |||
| JWH-176[10] | 碳氢 | 26 ± 4 | 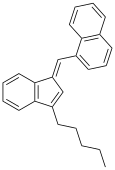 | |||
| JWH-180[5] | 萘甲酰基吲哚 | C25H25NO | 26 ± 2 | 9.6 ± 2.0 | CB2 (2.7x) | 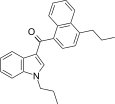 |
| JWH-181[5] | 萘甲酰基吲哚 | C28H31NO | 1.3 ± 0.1 | 0.62 ± 0.04 | CB2 (2.1x) | 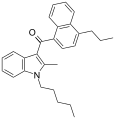 |
| JWH-182[5] | 萘甲酰基吲哚 | C27H29NO | 0.65 ± 0.03 | 1.1 ± 0.1 | CB1 (1.7x) | 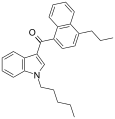 |
| JWH-184[10] | 萘甲基吲哚 | 23 ± 6 | 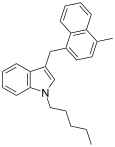 | |||
| JWH-185[10] | 萘甲基吲哚 | 17 ± 3 | 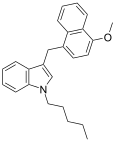 | |||
| JWH-186[14] | 二苯并吡喃 | 187 ± 23 | 5.6 ± 1.7 | CB2 (33x) | 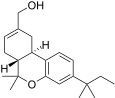 | |
| JWH-187[14] | 二苯并吡喃 | 84 ± 16 | 3.4 ± 0.5 | CB2 (25x) | 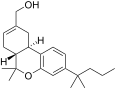 | |
| JWH-188[14] | 二苯并吡喃 | 270 ± 58 | 18 ± 2 | CB2 (15x) | 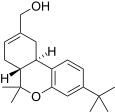 | |
| JWH-189[5] | 萘甲酰基吲哚 | C26H27NO | 52 ± 2 | 12 ± 0.8 | CB2 (4.3x) | 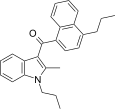 |
| JWH-190[14] | 二苯并吡喃 | 8.8 ± 1.4 | 1.6 ± 0.03 | CB2 (5.5x) | 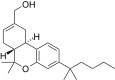 | |
| JWH-191[14] | 二苯并吡喃 | 1.8 ± 0.3 | 0.52 ± 0.03 | CB2 (3.5x) | 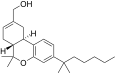 | |
| JWH-192[10] | 萘甲基吲哚 | 41 ± 13 | 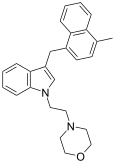 | |||
| JWH-193[10] | 萘甲酰基吲哚 | 6 ± 1 | 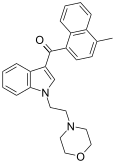 | |||
| JWH-194[10] | 萘甲基吲哚 | 127 ± 19 | 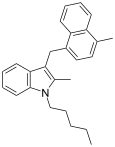 | |||
| JWH-195[10] | 萘甲基吲哚 | 113 ± 28 | 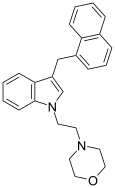 | |||
| JWH-196[10] | 萘甲基吲哚 | 151 ± 18 | 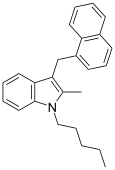 | |||
| JWH-197[10] | 萘甲基吲哚 | 323 ± 98 | 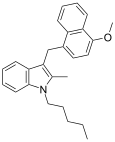 | |||
| JWH-198[10] | 萘甲酰基吲哚 | 10 ± 2 | 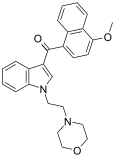 | |||
| JWH-199[10] | 萘甲基吲哚 | 20 ± 2 | 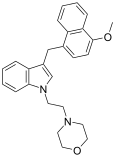 | |||
| JWH-200[10] | 萘甲酰基吲哚 | C25H24N2O2 | 42 ± 5 | 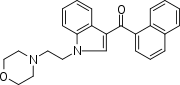 | ||
| JWH-201[15] | 苯乙酰吲哚 | C22H25NO2 | 1064 ± 21 | 444 ± 14 | CB2 (2.4x) | 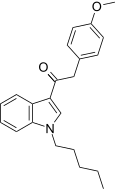 |
| JWH-202[15] | 苯乙酰吲哚 | C23H27NO2 | 1678 ± 63 | 645 ± 6 | CB2 (2.6x) | 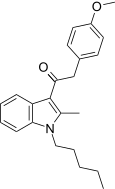 |
| JWH-203[15] | 苯乙酰吲哚 | 8.0 ± 0.9 | 7.0 ± 1.3 | — | 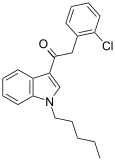 | |
| JWH-204[15] | 苯乙酰吲哚 | 13 ± 1 | 25 ± 1 | CB1 (1.9x) | 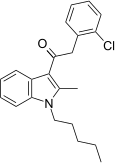 | |
| JWH-205[15] | 苯乙酰吲哚 | C22H25NO | 124 ± 23 | 180 ± 9 | CB1 (1.45x) | 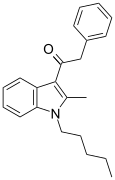 |
| JWH-206[15] | 苯乙酰吲哚 | 389 ± 25 | 498 ± 37 | CB1 (1.28x) | 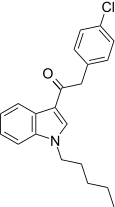 | |
| JWH-207[15] | 苯乙酰吲哚 | 1598 ± 134 | 3723 ± 10 | CB1 (2.33x) | 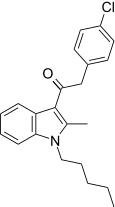 | |
| JWH-208[15] | 苯乙酰吲哚 | C22H25NO | 179 ± 7 | 570 ± 127 | CB1 (3.18x) | 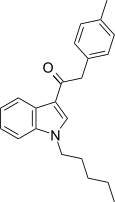 |
| JWH-209[15] | 苯乙酰吲哚 | C23H27NO | 746 ± 49 | 1353 ± 270 | CB1 (1.81x) | 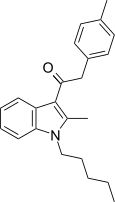 |
| JWH-210[5] | 萘甲酰基吲哚 | C26H27NO | 0.46 ± 0.03 | 0.69 ± 0.01 | CB1 (1.5x) | 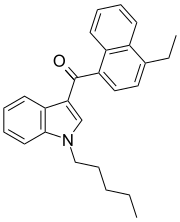 |
| JWH-211[5] | 萘甲酰基吲哚 | C25H25NO | 70 ± 0.8 | 12 ± 0.8 | CB2 (5.8x) | 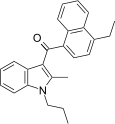 |
| JWH-212[5] | 萘甲酰基吲哚 | C24H23NO | 33 ± 0.9 | 10 ± 1.2 | CB2 (3.3x) | 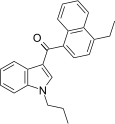 |
| JWH-213[5] | 萘甲酰基吲哚 | C27H29NO | 1.5 ± 0.2 | 0.42 ± 0.05 | CB2 (3.6x) | 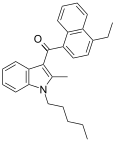 |
| JWH-215[14] | 二苯并吡喃 | 1008 ± 117 | 85 ± 21 | CB2 (12x) | 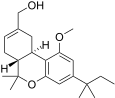 | |
| JWH-216[14] | 二苯并吡喃 | 1856 ± 148 | 333 ± 104 | CB2 (5.6x) | 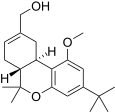 | |
| JWH-217[14] | 二苯并吡喃 | >10000 | 1404 ± 66 | CB2 (>7x) | 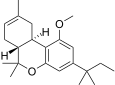 | |
| JWH-220 | 碳氢 | 19 | 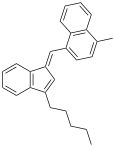 | |||
| JWH-224[14] | 二苯并吡喃 | 347 ± 34 | 28 ± 1 | CB2 (12.3x) | 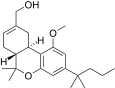 | |
| JWH-225[14] | 二苯并吡喃 | >10000 | 325 ± 70 | CB2 (>31x) | 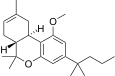 | |
| JWH-226[14] | 二苯并吡喃 | 4001 ± 282 | 43 ± 3 | CB2 (93x) | 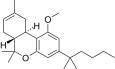 | |
| JWH-227[14] | 二苯并吡喃 | 40 ± 6 | 4.4 ± 0.3 | CB2 (9x) | 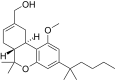 | |
| JWH-229[16] | 二苯并吡喃 | 3134 ± 110 | 18 ± 2 | CB2 (174x) | 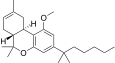 | |
| JWH-230[14] | 二苯并吡喃 | 15 ± 3 | 1.4 ± 0.12 | CB2 (10.7x) | 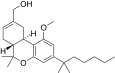 | |
| JWH-233[14] | 二苯并吡喃 | 14 ± 3 | 1.0 ± 0.3 | CB2 (14x) | 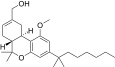 | |
| JWH-234[5] | 萘甲酰基吲哚 | C26H27NO | 8.4 ± 1.8 | 3.8 ± 0.6 | CB2 (2.2x) | 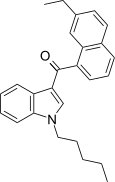 |
| JWH-235[5] | 萘甲酰基吲哚 | C24H23NO | 338 ± 34 | 123 ± 34 | CB2 (2.7x) | 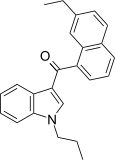 |
| JWH-236[5] | 萘甲酰基吲哚 | C25H25NO | 1351 ± 204 | 240 ± 63 | CB2 (5.6x) | 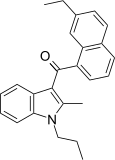 |
| JWH-237[15] | 苯乙酰吲哚 | 38 ± 10 | 106 ± 2 | CB1 (2.8x) | 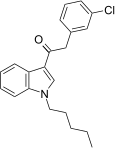 | |
| JWH-239[5] | 萘甲酰基吲哚 | C26H27NO | 342 ± 20 | 52 ± 6 | CB2 (6.6x) | 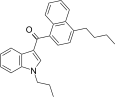 |
| JWH-240[5] | 萘甲酰基吲哚 | C28H31NO | 14 ± 1 | 7.2 ± 1.3 | CB2 (1.9x) | 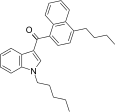 |
| JWH-241[5] | 萘甲酰基吲哚 | C27H29NO | 147 ± 20 | 49 ± 7 | CB2 (3.0x) | 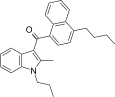 |
| JWH-242[5] | 萘甲酰基吲哚 | C29H33NO | 42 ± 9 | 6.5 ± 0.3 | CB2 (6.5x) | 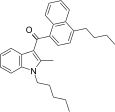 |
| JWH-243[13] | 萘甲酰基吡咯 | 285 ± 40 | 41 ± 3 | CB2 (6.95x) | 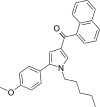 | |
| JWH-244[13] | 萘甲酰基吡咯 | 130 ± 6 | 18 ± 1 | CB2 (7.22x) | 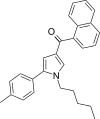 | |
| JWH-245[13] | 萘甲酰基吡咯 | 276 ± 4 | 25 ± 2 | CB2 (11x) | 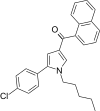 | |
| JWH-246[13] | 萘甲酰基吡咯 | 70 ± 4 | 16 ± 1 | CB2 (4.38x) | 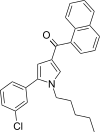 | |
| JWH-247[14] | 二苯并吡喃 | 427 ± 31 | 99 ± 4 | CB2 (4.3x) | 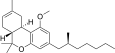 | |
| JWH-248[15] | 苯乙酰吲哚 | 1028 ± 39 | 657 ± 19 | CB2 (1.56x) | 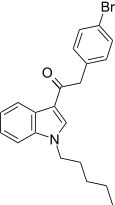 | |
| JWH-249[15] | 苯乙酰吲哚 | 8.4 ± 1.8 | 20 ± 2 | CB1 (2.38x) | 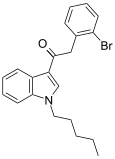 | |
| JWH-250[15] | 苯乙酰吲哚 | C22H25NO2 | 11 ± 2 | 33 ± 2 | CB1 (3x) | 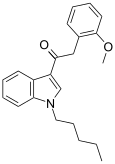 |
| JWH-251[15] | 苯乙酰吲哚 | C22H25NO | 29 ± 3 | 146 ± 36 | CB2 (5x) | 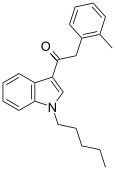 |
| JWH-252[15] | 苯乙酰吲哚 | C23H27NO | 23 ± 3 | 19 ± 1 | CB2 (1.2x) | 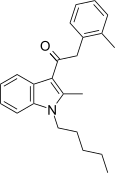 |
| JWH-253[15] | 苯乙酰吲哚 | C23H27NO2 | 62 ± 10 | 84 ± 12 | CB1 (1.35x) | 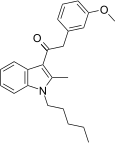 |
| JWH-254[14] | 二苯并吡喃 | 4724 ± 509 | 319 ± 16 | CB2 (14.8x) | 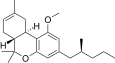 | |
| JWH-256[14] | 二苯并吡喃 | 4300 ± 888 | 97 ± 18 | CB2 (44x) | 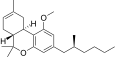 | |
| JWH-258[5] | 萘甲酰基吲哚 | C26H27NO2 | 4.6 ± 0.6 | 10.5 ± 1.3 | CB1 (2.3x) | 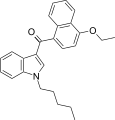 |
| JWH-259[5] | 萘甲酰基吲哚 | C24H23NO2 | 220 ± 29 | 74 ± 7 | CB2 (3.0x) | 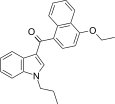 |
| JWH-260[5] | 萘甲酰基吲哚 | C27H29NO2 | 29 ± 0.4 | 25 ± 1.9 | CB2 (1.2x) | 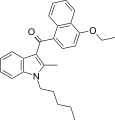 |
| JWH-261[5] | 萘甲酰基吲哚 | C25H25NO2 | 767 ± 105 | 221 ± 14 | CB2 (3.5x) | 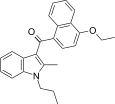 |
| JWH-262[5] | 萘甲酰基吲哚 | C27H29NO | 28 ± 3 | 5.6 ± 0.7 | CB2 (5.0x) | 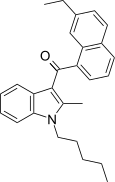 |
| JWH-265[5] | 萘甲酰基吲哚 | C23H21NO2 | 3788 ± 323 | 80 ± 13 | CB2 (47x) | 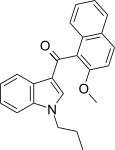 |
| JWH-266[5] | 萘甲酰基吲哚 | C24H23NO2 | >10000 | 455 ± 55 | CB2 (>22x) | 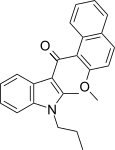 |
| JWH-267[5] | 萘甲酰基吲哚 | C25H25NO2 | 381 ± 16 | 7.2 ± 0.14 | CB2 (53x) |  |
| JWH-268[5] | 萘甲酰基吲哚 | C26H27NO2 | 1379 ± 193 | 40 ± 0.6 | CB2 (34x) | 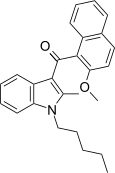 |
| JWH-277[14] | 二苯并吡喃 | 3905 ± 91 | 589 ± 65 | CB2 (6.6x) | 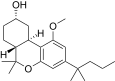 | |
| JWH-278[14] | 二苯并吡喃 | 906 ± 80 | 69 ± 6 | CB2 (13x) | 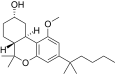 | |
| JWH-292[13] | 萘甲酰基吡咯 | 29 ± 1 | 20 ± 1 | CB2 (1.45x) | 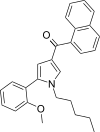 | |
| JWH-293[13] | 萘甲酰基吡咯 | 100 ± 5 | 41 ± 4 | CB2 (2.44x) | 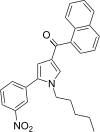 | |
| JWH-298[14] | 二苯并吡喃 | 812 ± 67 | 198 ± 23 | CB2 (4.1x) | 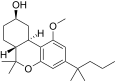 | |
| JWH-299[14] | 二苯并吡喃 | 415 ± 50 | 30 ± 2 | CB2 (13.8x) | 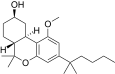 | |
| JWH-300[12] | 二苯并吡喃 | 118 ± 16 | 5.3 ± 0.1 | CB2 (22x) | 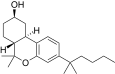 | |
| JWH-301[14] | 二苯并吡喃 | 295 ± 64 | 48 ± 4 | CB2 (6.1x) |  | |
| JWH-302[15] | 苯乙酰吲哚 | C22H25NO2 | 17 ± 2 | 89 ± 15 | CB1 (5.26x) | 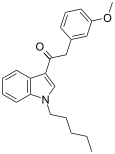 |
| JWH-303[15] | 苯乙酰吲哚 | 117 ± 10 | 138 ± 12 | CB1 (1.18x) | 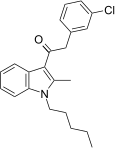 | |
| JWH-304[15] | 苯乙酰吲哚 | 3363 ± 332 | 2679 ± 688 | CB2 (1.26x) | 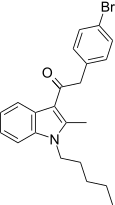 | |
| JWH-305[15] | 苯乙酰吲哚 | 15 ± 1.8 | 29 ± 5 | CB1 (1.93x) | 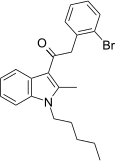 | |
| JWH-306[15] | 苯乙酰吲哚 | C23H27NO2 | 25 ± 1 | 82 ± 11 | CB1 (3.28x) | 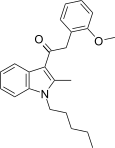 |
| JWH-307[13] | 萘甲酰基吡咯 | 7.7 ± 1.8 | 3.3 ± 0.2 | CB2 (2.33x) | 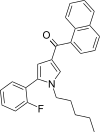 | |
| JWH-308[13] | 萘甲酰基吡咯 | 41 ± 1 | 33 ± 2 | CB2 (1.24x) | 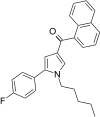 | |
| JWH-309[13] | 萘甲酰基吡咯 | 41 ± 3 | 49 ± 7 | CB1 (1.20x) | 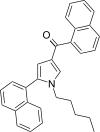 | |
| JWH-310[14] | 二苯并吡喃 | 1059 ± 51 | 36 ± 3 | CB2 (29x) | 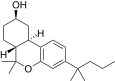 | |
| JWH-311[15] | 苯乙酰吲哚 | 23 ± 2 | 39 ± 3 | CB1 (1.70x) | 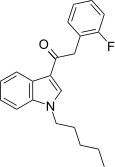 | |
| JWH-312[15] | 苯乙酰吲哚 | 72 ± 7 | 91 ± 20 | CB1 (1.26x) | 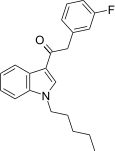 | |
| JWH-313[15] | 苯乙酰吲哚 | 422 ± 19 | 365 ± 92 | CB2 (1.16x) | 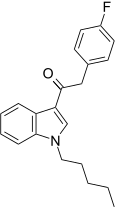 | |
| JWH-314[15] | 苯乙酰吲哚 | 39 ± 2 | 76 ± 4 | CB1 (1.95x) |  | |
| JWH-315[15] | 苯乙酰吲哚 | 430 ± 24 | 182 ± 23 | CB2 (3.36x) | 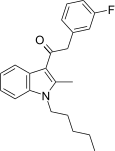 | |
| JWH-316[15] | 苯乙酰吲哚 | 2862 ± 670 | 781 ± 105 | CB2 (3.66x) | 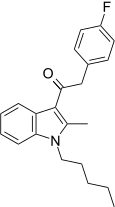 | |
| JWH-336[12] | 二苯并吡喃 | 4589 ± 367 | 153 ± 15 | CB2 (30x) | 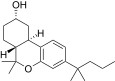 | |
| JWH-338[14] | 二苯并吡喃 | >10000 | 111 ± 16 | CB2 (>90x) | 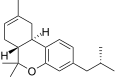 | |
| JWH-339[14] | 二苯并吡喃 | >10000 | 2317 ± 93 | CB2 (>4.3x) | 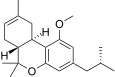 | |
| JWH-340[14] | 二苯并吡喃 | 135 ± 6 | 30 ± 1 | CB2 (4.5x) | 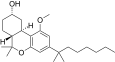 | |
| JWH-341[14] | 二苯并吡喃 | 100 ± 8 | 10 ± 0.1 | CB2 (10x) | 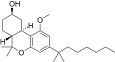 | |
| JWH-346[13] | 萘甲酰基吡咯 | 67 ± 6 | 39 ± 2 | CB2 (1.72x) | 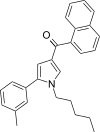 | |
| JWH-347[13] | 萘甲酰基吡咯 | 333 ± 17 | 169 ± 17 | CB2 (1.97x) | 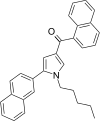 | |
| JWH-348[13] | 萘甲酰基吡咯 | 218 ± 19 | 53 ± 1 | CB2 (4.11x) | 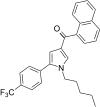 | |
| JWH-349[14] | 二苯并吡喃 | 376 ± 1 | 38 ± 4 | CB2 (9.9x) | 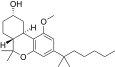 | |
| JWH-350[12] | 二苯并吡喃 | 395 ± 50 | 12 ± 1 | CB2 (33x) | 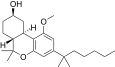 | |
| JWH-351[14] | 二苯并吡喃 | >10000 | 295 ± 3 | CB2 (>34x) | 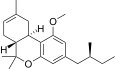 | |
| JWH-352[14] | 二苯并吡喃 | >10000 | 47 ± 2 | CB2 (>213x) | 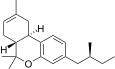 | |
| JWH-353[14] | 二苯并吡喃 | 1493 ± 10 | 31 ± 1 | CB2 (48x) | 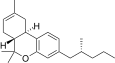 | |
| JWH-354[14] | 二苯并吡喃 | 1961 ± 21 | 241 ± 14 | CB2 (8.1x) | 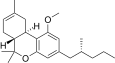 | |
| JWH-355[14] | 二苯并吡喃 | 2162 ± 220 | 108 ± 17 | CB2 (20x) | 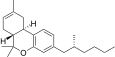 | |
| JWH-356[14] | 二苯并吡喃 | 5837 ± 701 | 108 ± 17 | CB2 (54x) | 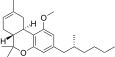 | |
| JWH-357[14] | 二苯并吡喃 | 647 ± 78 | 185 ± 4 | CB2 (3.5x) | 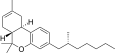 | |
| JWH-358[14] | 二苯并吡喃 | 1243 ± 266 | 52 ± 3 | CB2 (24x) | 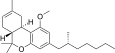 | |
| JWH-359 | 二苯并吡喃 | C24H36O2 | 2918 ± 450 | 13.0 ± 0.2 | CB2 (220x) | 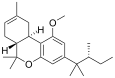 |
| JWH-360[14] | 二苯并吡喃 | 2449 ± 606 | 160 ± 8 | CB2 (15x) | 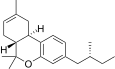 | |
| JWH-361[14] | 二苯并吡喃 | 63 ± 3 | 2.7 ± 0.1 | CB2 (23x) | 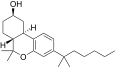 | |
| JWH-362[14] | 二苯并吡喃 | 127 ± 8 | 34 ± 5 | CB2 (3.7x) |  | |
| JWH-363[13] | 萘甲酰基吡咯 | 245 ± 5 | 71 ± 1 | CB2 (3.45x) | 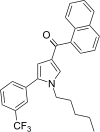 | |
| JWH-364[13] | 萘甲酰基吡咯 | C28H29NO | 34 ± 3 | 29 ± 1 | CB2 (1.17x) | 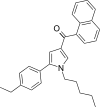 |
| JWH-365[13] | 萘甲酰基吡咯 | C28H29NO | 17 ± 1 | 3.4 ± 0.2 | CB2 (5.0x) | 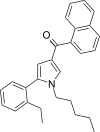 |
| JWH-366[13] | 萘甲酰基吡咯 | C25H24N2O | 191 ± 12 | 24 ± 1 | CB2 (7.96x) | 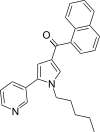 |
| JWH-367[13] | 萘甲酰基吡咯 | C27H27NO2 | 53 ± 2 | 23 ± 1 | CB2 (2.30x) | 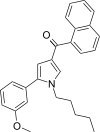 |
| JWH-368[13] | 萘甲酰基吡咯 | 16 ± 1 | 9.1 ± 0.7 | CB2 (1.76x) | 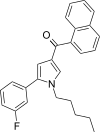 | |
| JWH-369[13] | 萘甲酰基吡咯 | 7.9 ± 0.4 | 5.2 ± 0.3 | CB2 (1.52x) | 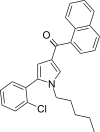 | |
| JWH-370[13] | 萘甲酰基吡咯 | C27H27NO | 5.6 ± 0.4 | 4.0 ± 0.5 | CB2 (1.40x) | 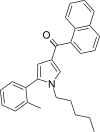 |
| JWH-371[13] | 萘甲酰基吡咯 | C30H33NO | 42 ± 1 | 64 ± 2 | CB1 (1.52x) | 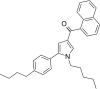 |
| JWH-372[13] | 萘甲酰基吡咯 | 77 ± 2 | 8.2 ± 0.2 | CB1 (9.39x) | 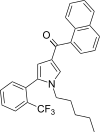 | |
| JWH-373[13] | 萘甲酰基吡咯 | C30H33NO | 60 ± 3 | 69 ± 2 | CB1 (1.15x) | 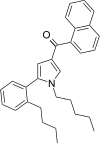 |
| JWH-387[17] | 萘甲酰基吲哚 | C24H22BrNO | 1.2 ± 0.1 | 1.1 ± 0.1 | — | 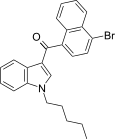 |
| JWH-398[18] | 萘甲酰基吲哚 | 2.3 ± 0.1 | 2.8 ± 0.2 | CB1 (1.22x) | ||
| JWH-416[17] | 萘甲酰基吲哚 | 73 ± 10 | 3.3 ± 0.1 | CB2 (22x) | 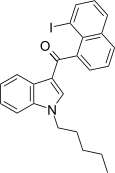 | |
| JWH-417[17] | 萘甲酰基吲哚 | 522 ± 58 | 13 ± 0.2 | CB2 (40x) | 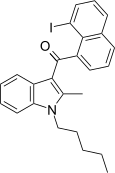 | |
| JWH-422[17] | 萘甲酰基吲哚 | 501 ± 48 | 20 ± 0.4 | CB2 (25x) | 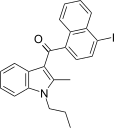 | |
| JWH-423[17] | 萘甲酰基吲哚 | 140 ± 10 | 6.6 ± 0.2 | CB2 (21x) | 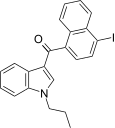 | |
| JWH-424[17] | 萘甲酰基吲哚 | C24H22BrNO | 21 ± 3.4 | 5.4 ± 0.2 | CB2 (3.9x) | 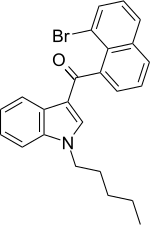 |
| JWH-425[17] | 萘甲酰基吲哚 | C25H24BrNO | 54 ± 11 | 10 ± 0.4 | CB2 (5.4x) | 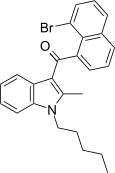 |
参见
- AM大麻素列表
- CP大麻素列表
- HU大麻素列表
- 杂项设计师大麻素列表
注释
- Ki是化合物对大麻素1型受体(CB1)或大麻素2型受体(CB2)的结合亲和力
参考资料
- Manera C, Tuccinardi T, Martinelli A. . Mini Rev Med Chem. 2008, 8 (4): 370–87. PMID 18473928. doi:10.2174/138955708783955935.
- Wiley JL, Marusich JA, Huffman JW. . Life Sci. 2014, 97 (1): 55–63. PMC 3944940
 . PMID 24071522. doi:10.1016/j.lfs.2013.09.011.
. PMID 24071522. doi:10.1016/j.lfs.2013.09.011. - Wiley JL, Marusich JA, Thomas BF. . Curr Top Behav Neurosci. Current Topics in Behavioral Neurosciences. 2017, 32: 231–248. ISBN 978-3-319-52442-9. PMID 27753007. doi:10.1007/7854_2016_17
 .
. - Banister SD, Connor M. . Handb Exp Pharmacol. Handbook of Experimental Pharmacology. 2018, 252: 165–190. ISBN 978-3-030-10560-0. PMID 29980914. doi:10.1007/164_2018_143.
- Huffman JW, Zengin G, Wu MJ, Lu J, Hynd G, Bushell K, Thompson AL, Bushell S, Tartal C, Hurst DP, Reggio PH, Selley DE, Cassidy MP, Wiley JL, Martin BR. . Bioorganic & Medicinal Chemistry. January 2005, 13 (1): 89–112. PMID 15582455. doi:10.1016/j.bmc.2004.09.050.
- Aung MM, Griffin G, Huffman JW, Wu M, Keel C, Yang B, Showalter VM, Abood ME, Martin BR. . Drug and Alcohol Dependence. August 2000, 60 (2): 133–40. PMID 10940540. doi:10.1016/S0376-8716(99)00152-0.
- Huffman JW, Liddle J, Yu S, Aung MM, Abood ME, Wiley JL, Martin BR. . Bioorganic & Medicinal Chemistry. December 1999, 7 (12): 2905–14. PMID 10658595. doi:10.1016/s0968-0896(99)00219-9.
- Huffman JW, Yu S, Showalter V, Abood ME, Wiley JL, Compton DR, Martin BR, Bramblett RD, Reggio PH. . Journal of Medicinal Chemistry. September 1996, 39 (20): 3875–7. PMID 8831752. doi:10.1021/JM960394Y.
- Bow EW, Rimoldi JM. The Structure-Function Relationships of Classical Cannabinoids: CB1/CB2 Modulation. Perspect Medicin Chem. 2016 Jun 28;8:17-39. doi:10.4137/PMC.S32171 PMID 27398024
- Huffman JW, Mabon R, Wu MJ, Lu J, Hart R, Hurst DP, Reggio PH, Wiley JL, Martin BR. . Bioorganic & Medicinal Chemistry. February 2003, 11 (4): 539–49. PMID 12538019. S2CID 29107765. doi:10.1016/S0968-0896(02)00451-0.
- Martin BR, Jefferson R, Winckler R, Wiley JL, Huffman JW, Crocker PJ, Saha B, Razdan RK. Manipulation of the tetrahydrocannabinol side chain delineates agonists, partial agonists, and antagonists. J Pharmacol Exp Ther. 1999 Sep;290(3):1065-79. PMID 10454479
- Howlett AC, Barth F, Bonner TI, Cabral G, Casellas P, Devane WA, Felder CC, Herkenham M, Mackie K, Martin BR, Mechoulam R, Pertwee RG. . Pharmacological Reviews. June 2002, 54 (2): 161–202. PMID 12037135. S2CID 8259002. doi:10.1124/pr.54.2.161.
- Huffman JW, Padgett LW, Isherwood ML, Wiley JL, Martin BR. . Bioorganic & Medicinal Chemistry Letters. October 2006, 16 (20): 5432–5. PMID 16889960. doi:10.1016/j.bmcl.2006.07.051.
- Marriott KS, Huffman JW. . Current Topics in Medicinal Chemistry. 2008, 8 (3): 187–204. PMID 18289088. doi:10.2174/156802608783498014.
- Huffman JW, Szklennik PV, Almond A, Bushell K, Selley DE, He H, Cassidy MP, Wiley JL, Martin BR. . Bioorganic & Medicinal Chemistry Letters. September 2005, 15 (18): 4110–3. PMID 16005223. doi:10.1016/j.bmcl.2005.06.008.
- Huffman JW, Bushell SM, Miller JR, Wiley JL, Martin BR. . Bioorganic & Medicinal Chemistry. December 2002, 10 (12): 4119–29. PMID 12413866. doi:10.1016/s0968-0896(02)00331-0.
- Wiley JL, Smith VJ, Chen J, Martin BR, Huffman JW. . Bioorganic & Medicinal Chemistry. 2012, 20 (6): 2067–2081. PMC 3298571
 . PMID 22341572. doi:10.1016/j.bmc.2012.01.038.
. PMID 22341572. doi:10.1016/j.bmc.2012.01.038. - . The Receptors. 2009. ISBN 978-1-58829-712-9. doi:10.1007/978-1-59745-503-9.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.