 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| Chemical and physical data | |
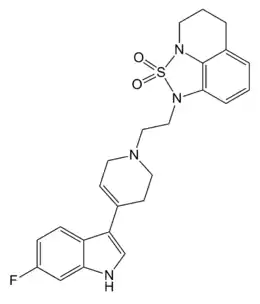
| Formula | C24H25FN4O2S |
| Molar mass | 452.55 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
LY-367,265 is a drug developed by Eli Lilly, which acts as both a potent and selective antagonist at the serotonin 5-HT2A receptor, and also a selective serotonin reuptake inhibitor (SSRI). It has antidepressant effects in animal studies, reduces glutamate signalling in the brain and increases the analgesic effects of morphine.[1][2][3]
References
- ↑ Pullar IA, Carney SL, Colvin EM, Lucaites VL, Nelson DL, Wedley S (October 2000). "LY367265, an inhibitor of the 5-hydroxytryptamine transporter and 5-hydroxytryptamine(2A) receptor antagonist: a comparison with the antidepressant, nefazodone". European Journal of Pharmacology. 407 (1–2): 39–46. doi:10.1016/S0014-2999(00)00728-7. PMID 11050288.
- ↑ Wang SJ (March 2005). "Potential antidepressant LY 367265 presynaptically inhibits the release of glutamate in rat cerebral cortex". Synapse. 55 (3): 156–63. doi:10.1002/syn.20104. PMID 15602751. S2CID 11860623.
- ↑ Ozdemir E, Bagcivan I, Gursoy S, Altun A, Durmus N (June 2011). "Effects of fluoxetine and LY 367265 on tolerance to the analgesic effect of morphine in rats". Acta Physiologica Hungarica. 98 (2): 205–13. doi:10.1556/APhysiol.98.2011.2.12. PMID 21616779.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.