 | |
 | |
| Names | |
|---|---|
| IUPAC name
Aminourea[1] | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.308 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
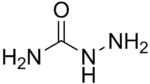
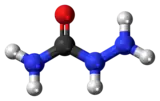
| H2NNHC(=O)NH2 | |
| Molar mass | 75.08 g/mol |
| Melting point | 96 °C |
| Hazards | |
| GHS labelling: | |
  | |
| Danger | |
| H301, H315, H319, H335 | |
| P261, P264, P270, P271, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P312, P330, P332+P313, P337+P313, P362, P403+P233, P405 | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Semicarbazide is the chemical compound with the formula OC(NH2)(N2H3). It is a water-soluble white solid. It is a derivative of urea.
Synthesis
The compound prepared by treating urea with hydrazine:[2]
- OC(NH2)2 + N2H4 → OC(NH2)(N2H3) + NH3
A further reaction can occur to give carbohydrazide:
- OC(NH2)(N2H3) + N2H4 → OC(N2H3)2 + NH3
Derivatives
Semicarbazide is frequently reacted with aldehydes and ketones to produce semicarbazones via a condensation reaction. This is an example of imine formation resulting from the reaction of a primary amine with a carbonyl group. The reaction is useful because semicarbazones, like oximes and 2,4-DNPs, typically have high melting points and crystallize, facilitating purification or identification of reaction products.[3]
Properties
Semicarbazide products (semicarbazones and thiosemicarbazones) are known to have an activity of antiviral, antiinfective and antineoplastic through binding to copper or iron in cells.
Uses, occurrence, detection
Semicarbazide is used in preparing pharmaceuticals including nitrofuran antibacterials (furazolidone, nitrofurazone, nitrofurantoin) and related compounds. It is also a product of degradations of the blowing agent azodicarbonamide (ADC). Semicarbazide forms in heat-treated flour containing ADC as well as breads made from ADC-treated flour.[4][5]
Semicarbazide is used as a detection reagent in thin layer chromatography (TLC). Semicarbazide stains α-keto acids on the TLC plate, which can then be viewed under ultraviolet light.
See also
- Biurea - another product of reaction of urea with hydrazine
- Carbazide - structurally related with the general formula (R2NNH)2C(O)
- Semicarbazide-cadmium therapy
- thiosemicarbazide
References
- ↑ CID 5196 from PubChem
- ↑ Jean-Pierre Schirmann, Paul Bourdauducq "Hydrazine" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2002. doi:10.1002/14356007.a13_177.
- ↑ John McMurry (1984). Organic Chemistry. Brooks/Cole. p. 676.
- ↑ Becalski, Adam; Lau, Benjamin; Lewis, David; Seaman, Stephen (2004). "Semicarbazide Formation in Azodicarbonamide-Treated Flour: A Model Study". J. Agric. Food Chem. 52 (18): 5730–4. doi:10.1021/jf0495385. PMID 15373416.
- ↑ Maria Beatriz de la Calle; Elke Anklam (2005). "Semicarbazide: occurrence in food products and state-of-the-art in analytical methods used for its determination". Anal. Bioanal. Chem. 382 (4): 968–977. doi:10.1007/s00216-005-3243-z. PMID 15947918. S2CID 10765283.
