| UBA1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | UBA1, A1S9, A1S9T, A1ST, AMCX1, CFAP124, GXP1, POC20, SMAX2, UBA1A, UBE1, UBE1X, ubiquitin like modifier activating enzyme 1, VEXAS | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 314370 MGI: 98890 HomoloGene: 22002 GeneCards: UBA1 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Ubiquitin-like modifier activating enzyme 1 (UBA1) is an enzyme which in humans is encoded by the UBA1 gene.[5][6] UBA1 participates in ubiquitination and the NEDD8 pathway for protein folding and degradation, among many other biological processes.[5][7] This protein has been linked to X-linked spinal muscular atrophy type 2, neurodegenerative diseases, and cancers.[8][9]
Structure
Gene
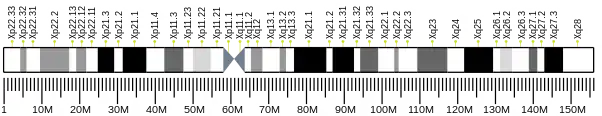
The UBA1 gene is located in the chromosome band Xp11.23, consisting of 31 exons.
Protein
The UBA1 for ubiquitin (Ub) is a 110–120 kDa monomeric protein, and the UBA1 for the ubiquitin-like protein (Ubls) NEDD8 and SUMO are heterodimeric complexes with similar molecular weights. All eukaryotic UBA1 contain a two-fold repeat of a domain, derived from the bacterial MoeB and ThiF proteins,[10] with one occurrence each in the N-terminal and C-terminal half of the UBA1 for Ub, or the separate subunits of the UBA1 for NEDD8 and SUMO.[11] The UBA1 for Ub consists of four building blocks: First, the adenylation domains composed of two MoeB/ThiF-homology motifs, the latter of which binds ATP and Ub;[12][13][14] second, the catalytic cysteine half-domains, which contain the E1 active site cysteine inserted into each of the adenylation domains;[15] third, a four-helix bundle that represents a second insertion in the inactive adenylation domain and immediately follows the first catalytic cysteine half-domain; and fourth, the C-terminal ubiquitin-fold domain, which recruits specific E2s.[13][16][17]
Function
The protein encoded by this gene catalyzes the first step in ubiquitin conjugation, or ubiquitination, to mark cellular proteins for degradation. Specifically, UBA1 catalyzes the ATP-dependent adenylation of ubiquitin, thereby forming a thioester bond between the two. It also continues to participate in subsequent steps of ubiquination as a Ub carrier.[8][9][18] There are only two human ubiquitin-activating enzymes, UBA1 and UBA6, and thus UBA1 is largely responsible for protein ubiquitination in humans.[8][9][18] Through its central role in ubiquitination, UBA1 has been linked to cell cycle regulation, endocytosis, signal transduction, apoptosis, DNA damage repair, and transcriptional regulation.[8][9] Additionally, UBA1 helps regulate the NEDD8 pathway, thus implicating it in protein folding, as well as mitigating the depletion of ubiquitin levels during stress.[7]
Clinical significance
Mutations in UBA1 are associated with X-linked spinal muscular atrophy type 2.[5] UBA1 has also been implicated in other neurodegenerative diseases, including spinal muscular atrophy,[19] as well as cancer and tumors. Since UBA1 is involved in multiple biological processes, there are concerns that inhibiting UBA1 would also damage normal cells. Nonetheless, preclinical testing of a UBA1 inhibitor in mice with leukemia revealed no additional toxic effects to normal cells, and the success of other drugs targeting pleiotropic targets likewise support the safety of using UBA1 inhibitor in cancer treatment[8][9]
Moreover, the UBA1 inhibitor largazole, as well as its ketone and ester derivatives, preferentially targets cancer over normal cells by specifically blocking the ligation of Ub and UBA1 during the adenylation step of the E1 pathway. MLN4924, a NEDD8-activating enzyme inhibitor functioning according to similar mechanisms, is currently undergoing phase I clinical trials.[9]
An autoinflammatory condition identified in 2020 and named VEXAS syndrome (vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic) is due to mutation in methionine41 in UBA1, the E1 enzyme that initiates ubiquitylation.[20]
Interactions
UBA1 has been shown to interact with:
References
- 1 2 3 GRCh38: Ensembl release 89: ENSG00000130985 - Ensembl, May 2017
- 1 2 3 GRCm38: Ensembl release 89: ENSMUSG00000001924 - Ensembl, May 2017
- ↑ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- 1 2 3 "Entrez Gene: ubiquitin-like modifier activating enzyme 1".
- ↑ Kudo M, Sugasawa K, Hori T, Enomoto T, Hanaoka F, Ui M (January 1991). "Human ubiquitin-activating enzyme (E1): compensation for heat-labile mouse E1 and its gene localization on the X chromosome". Experimental Cell Research. 192 (1): 110–7. doi:10.1016/0014-4827(91)90164-P. PMID 1845793.
- 1 2 Leidecker O, Matic I, Mahata B, Pion E, Xirodimas DP (March 2012). "The ubiquitin E1 enzyme Ube1 mediates NEDD8 activation under diverse stress conditions". Cell Cycle. 11 (6): 1142–50. doi:10.4161/cc.11.6.19559. PMID 22370482.
- 1 2 3 4 5 6 Correale S, de Paola I, Morgillo CM, Federico A, Zaccaro L, Pallante P, Galeone A, Fusco A, Pedone E, Luque FJ, Catalanotti B (2014). "Structural model of the hUbA1-UbcH10 quaternary complex: in silico and experimental analysis of the protein-protein interactions between E1, E2 and ubiquitin". PLOS ONE. 9 (11): e112082. Bibcode:2014PLoSO...9k2082C. doi:10.1371/journal.pone.0112082. PMC 4223017. PMID 25375166.
- 1 2 3 4 5 6 Ungermannova D, Parker SJ, Nasveschuk CG, Wang W, Quade B, Zhang G, Kuchta RD, Phillips AJ, Liu X (2012). "Largazole and its derivatives selectively inhibit ubiquitin activating enzyme (e1)". PLOS ONE. 7 (1): e29208. Bibcode:2012PLoSO...729208U. doi:10.1371/journal.pone.0029208. PMC 3261141. PMID 22279528.
- ↑ Johnson ES, Schwienhorst I, Dohmen RJ, Blobel G (September 1997). "The ubiquitin-like protein Smt3p is activated for conjugation to other proteins by an Aos1p/Uba2p heterodimer". The EMBO Journal. 16 (18): 5509–19. doi:10.1093/emboj/16.18.5509. PMC 1170183. PMID 9312010.
- ↑ Lee I, Schindelin H (July 2008). "Structural insights into E1-catalyzed ubiquitin activation and transfer to conjugating enzymes". Cell. 134 (2): 268–78. doi:10.1016/j.cell.2008.05.046. PMID 18662542.
- ↑ Lake MW, Wuebbens MM, Rajagopalan KV, Schindelin H (November 2001). "Mechanism of ubiquitin activation revealed by the structure of a bacterial MoeB-MoaD complex" (PDF). Nature. 414 (6861): 325–9. Bibcode:2001Natur.414..325L. doi:10.1038/35104586. PMID 11713534. S2CID 3224437.
- 1 2 Lois LM, Lima CD (February 2005). "Structures of the SUMO E1 provide mechanistic insights into SUMO activation and E2 recruitment to E1". The EMBO Journal. 24 (3): 439–51. doi:10.1038/sj.emboj.7600552. PMC 548657. PMID 15660128.
- ↑ Walden H, Podgorski MS, Schulman BA (March 2003). "Insights into the ubiquitin transfer cascade from the structure of the activating enzyme for NEDD8". Nature. 422 (6929): 330–4. Bibcode:2003Natur.422..330W. doi:10.1038/nature01456. PMID 12646924. S2CID 4370095.
- ↑ Szczepanowski RH, Filipek R, Bochtler M (June 2005). "Crystal structure of a fragment of mouse ubiquitin-activating enzyme". The Journal of Biological Chemistry. 280 (23): 22006–11. doi:10.1074/jbc.M502583200. PMID 15774460.
- ↑ Huang DT, Paydar A, Zhuang M, Waddell MB, Holton JM, Schulman BA (February 2005). "Structural basis for recruitment of Ubc12 by an E2 binding domain in NEDD8's E1". Molecular Cell. 17 (3): 341–50. doi:10.1016/j.molcel.2004.12.020. PMID 15694336.
- ↑ Huang DT, Hunt HW, Zhuang M, Ohi MD, Holton JM, Schulman BA (January 2007). "Basis for a ubiquitin-like protein thioester switch toggling E1-E2 affinity". Nature. 445 (7126): 394–8. Bibcode:2007Natur.445..394H. doi:10.1038/nature05490. PMC 2821831. PMID 17220875.
- 1 2 Moudry P, Lukas C, Macurek L, Hanzlikova H, Hodny Z, Lukas J, Bartek J (April 2012). "Ubiquitin-activating enzyme UBA1 is required for cellular response to DNA damage". Cell Cycle. 11 (8): 1573–82. doi:10.4161/cc.19978. PMID 22456334.
- ↑ Powis, Rachael A.; Karyka, Evangelia; Boyd, Penelope; Côme, Julien; Jones, Ross A.; Zheng, Yinan; Szunyogova, Eva; Groen, Ewout J.N.; Hunter, Gillian; Thomson, Derek; Wishart, Thomas M.; Becker, Catherina G.; Parson, Simon H.; Martinat, Cécile; Azzouz, Mimoun; Gillingwater, Thomas H. (2016). "Systemic restoration of UBA1 ameliorates disease in spinal muscular atrophy" (PDF). JCI Insight. 1 (11): e87908. doi:10.1172/jci.insight.87908. PMC 5033939. PMID 27699224.
- ↑ Beck, David B.; Ferrada, Marcela A.; Sikora, Keith A.; Ombrello, Amanda K.; Collins, Jason C.; Pei, Wuhong; Balanda, Nicholas; Ross, Daron L.; Ospina Cardona, Daniela; Wu, Zhijie; Patel, Bhavisha (2020-10-27). "Somatic Mutations in UBA1 and Severe Adult-Onset Autoinflammatory Disease". New England Journal of Medicine. 383 (27): 2628–2638. doi:10.1056/NEJMoa2026834. ISSN 0028-4793. PMC 7847551. PMID 33108101.
- ↑ Qin Z, Cui B, Jin J, Song M, Zhou B, Guo H, Qian D, He Y, Huang L (April 2016). "The ubiquitin-activating enzyme E1 as a novel therapeutic target for the treatment of restenosis". Atherosclerosis. 247: 142–53. doi:10.1016/j.atherosclerosis.2016.02.016. PMID 26919560.
- ↑ Tsukamoto S (2016). "Search for Inhibitors of the Ubiquitin-Proteasome System from Natural Sources for Cancer Therapy". Chemical & Pharmaceutical Bulletin. 64 (2): 112–8. doi:10.1248/cpb.c15-00768. PMID 26833439.
- ↑ Yamanokuchi R, Imada K, Miyazaki M, Kato H, Watanabe T, Fujimuro M, Saeki Y, Yoshinaga S, Terasawa H, Iwasaki N, Rotinsulu H, Losung F, Mangindaan RE, Namikoshi M, de Voogd NJ, Yokosawa H, Tsukamoto S (July 2012). "Hyrtioreticulins A-E, indole alkaloids inhibiting the ubiquitin-activating enzyme, from the marine sponge Hyrtios reticulatus". Bioorganic & Medicinal Chemistry. 20 (14): 4437–42. doi:10.1016/j.bmc.2012.05.044. PMID 22695182.
Further reading
- Nouspikel T, Hanawalt PC (October 2006). "Impaired nucleotide excision repair upon macrophage differentiation is corrected by E1 ubiquitin-activating enzyme". Proceedings of the National Academy of Sciences of the United States of America. 103 (44): 16188–93. Bibcode:2006PNAS..10316188N. doi:10.1073/pnas.0607769103. PMC 1621053. PMID 17060614.
- Jin J, Li X, Gygi SP, Harper JW (June 2007). "Dual E1 activation systems for ubiquitin differentially regulate E2 enzyme charging". Nature. 447 (7148): 1135–8. Bibcode:2007Natur.447.1135J. doi:10.1038/nature05902. PMID 17597759. S2CID 4337767.
- Xia T, Dimitropoulou C, Zeng J, Antonova GN, Snead C, Venema RC, Fulton D, Qian S, Patterson C, Papapetropoulos A, Catravas JD (November 2007). "Chaperone-dependent E3 ligase CHIP ubiquitinates and mediates proteasomal degradation of soluble guanylyl cyclase". American Journal of Physiology. Heart and Circulatory Physiology. 293 (5): H3080–7. doi:10.1152/ajpheart.00579.2007. PMID 17873020.
- Pridgeon JW, Webber EA, Sha D, Li L, Chin LS (January 2009). "Proteomic analysis reveals Hrs ubiquitin-interacting motif-mediated ubiquitin signaling in multiple cellular processes". The FEBS Journal. 276 (1): 118–31. doi:10.1111/j.1742-4658.2008.06760.x. PMC 2647816. PMID 19019082.
- Beausoleil SA, Villén J, Gerber SA, Rush J, Gygi SP (October 2006). "A probability-based approach for high-throughput protein phosphorylation analysis and site localization". Nature Biotechnology. 24 (10): 1285–92. doi:10.1038/nbt1240. PMID 16964243. S2CID 14294292.
- Malakhova OA, Zhang DE (April 2008). "ISG15 inhibits Nedd4 ubiquitin E3 activity and enhances the innate antiviral response". The Journal of Biological Chemistry. 283 (14): 8783–7. doi:10.1074/jbc.C800030200. PMC 2276364. PMID 18287095.
- Anindya R, Aygün O, Svejstrup JQ (November 2007). "Damage-induced ubiquitylation of human RNA polymerase II by the ubiquitin ligase Nedd4, but not Cockayne syndrome proteins or BRCA1". Molecular Cell. 28 (3): 386–97. doi:10.1016/j.molcel.2007.10.008. PMID 17996703.
- Umebayashi K, Stenmark H, Yoshimori T (August 2008). "Ubc4/5 and c-Cbl continue to ubiquitinate EGF receptor after internalization to facilitate polyubiquitination and degradation". Molecular Biology of the Cell. 19 (8): 3454–62. doi:10.1091/mbc.E07-10-0988. PMC 2488299. PMID 18508924.
- Barbe L, Lundberg E, Oksvold P, Stenius A, Lewin E, Björling E, Asplund A, Pontén F, Brismar H, Uhlén M, Andersson-Svahn H (March 2008). "Toward a confocal subcellular atlas of the human proteome". Molecular & Cellular Proteomics. 7 (3): 499–508. doi:10.1074/mcp.M700325-MCP200. PMID 18029348.
- Lim J, Hao T, Shaw C, Patel AJ, Szabó G, Rual JF, Fisk CJ, Li N, Smolyar A, Hill DE, Barabási AL, Vidal M, Zoghbi HY (May 2006). "A protein-protein interaction network for human inherited ataxias and disorders of Purkinje cell degeneration". Cell. 125 (4): 801–14. doi:10.1016/j.cell.2006.03.032. PMID 16713569.
- Gallegos JR, Litersky J, Lee H, Sun Y, Nakayama K, Nakayama K, Lu H (January 2008). "SCF TrCP1 activates and ubiquitylates TAp63gamma". The Journal of Biological Chemistry. 283 (1): 66–75. doi:10.1074/jbc.M704686200. PMID 17965458.
- Carbia-Nagashima A, Gerez J, Perez-Castro C, Paez-Pereda M, Silberstein S, Stalla GK, Holsboer F, Arzt E (October 2007). "RSUME, a small RWD-containing protein, enhances SUMO conjugation and stabilizes HIF-1alpha during hypoxia". Cell. 131 (2): 309–23. doi:10.1016/j.cell.2007.07.044. hdl:20.500.12110/paper_00928674_v131_n2_p309_CarbiaNagashima. PMID 17956732. S2CID 12486303.
- Ramser J, Ahearn ME, Lenski C, Yariz KO, Hellebrand H, von Rhein M, Clark RD, Schmutzler RK, Lichtner P, Hoffman EP, Meindl A, Baumbach-Reardon L (January 2008). "Rare missense and synonymous variants in UBE1 are associated with X-linked infantile spinal muscular atrophy". American Journal of Human Genetics. 82 (1): 188–93. doi:10.1016/j.ajhg.2007.09.009. PMC 2253959. PMID 18179898.
- Zenke-Kawasaki Y, Dohi Y, Katoh Y, Ikura T, Ikura M, Asahara T, Tokunaga F, Iwai K, Igarashi K (October 2007). "Heme induces ubiquitination and degradation of the transcription factor Bach1". Molecular and Cellular Biology. 27 (19): 6962–71. doi:10.1128/MCB.02415-06. PMC 2099246. PMID 17682061.
- Nicassio F, Corrado N, Vissers JH, Areces LB, Bergink S, Marteijn JA, Geverts B, Houtsmuller AB, Vermeulen W, Di Fiore PP, Citterio E (November 2007). "Human USP3 is a chromatin modifier required for S phase progression and genome stability". Current Biology. 17 (22): 1972–7. doi:10.1016/j.cub.2007.10.034. PMID 17980597.
- Su ZL, Mo XL, Feng ZY, Lin HL, Ding YG (September 2008). "UBE1 expression in extranodal NK/T cell lymphoma, nasal type". Leukemia & Lymphoma. 49 (9): 1821–2. doi:10.1080/10428190802187171. PMID 18661401. S2CID 5481217.
- Wang X, Shi Y, Wang J, Huang G, Jiang X (September 2008). "Crucial role of the C-terminus of PTEN in antagonizing NEDD4-1-mediated PTEN ubiquitination and degradation". The Biochemical Journal. 414 (2): 221–9. doi:10.1042/BJ20080674. PMID 18498243.
- Bruce MC, Kanelis V, Fouladkou F, Debonneville A, Staub O, Rotin D (October 2008). "Regulation of Nedd4-2 self-ubiquitination and stability by a PY motif located within its HECT-domain". The Biochemical Journal. 415 (1): 155–63. doi:10.1042/BJ20071708. PMID 18498246.
- Zhou W, Zhu P, Wang J, Pascual G, Ohgi KA, Lozach J, Glass CK, Rosenfeld MG (January 2008). "Histone H2A monoubiquitination represses transcription by inhibiting RNA polymerase II transcriptional elongation". Molecular Cell. 29 (1): 69–80. doi:10.1016/j.molcel.2007.11.002. PMC 2327256. PMID 18206970.
External links
- UBE1+Protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
This article incorporates text from the United States National Library of Medicine, which is in the public domain.