5-脱氢表甾醇
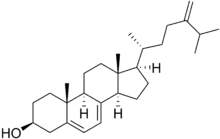
5-脱氢表甾醇(英語:,化学式C28H44O)是油菜素類固醇等物质生物合成过程中的中间产物[1],由C-5甾醇去饱和酶催化表甾醇生成[2],再通过7-脱氢胆固醇还原酶转化为24-亚甲基胆固醇[3]。
| 5-脱氢表甾醇 | |
|---|---|
 | |
 | |
| IUPAC名 (3S,10R,13R)-17-[(1R)-1,5-dimethyl-4-methylenehexyl]-10,13-dimethyl-2,3,4,9,11,12,14,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-3-ol | |
| 别名 | 24-Methylcholesta-5,7,24(28)-trienol, ergosta-5,7,24(28)-trien-3β-ol, campesta-7,24(28)-dien-3β-ol |
| 识别 | |
| CAS号 | 23582-83-4 |
| PubChem | 10894570 |
| ChemSpider | 9069833 |
| SMILES |
|
| InChI |
|
| InChIKey | ZEPNVCGPJXYABB-LOIOQLKMBD |
| KEGG | C15780 |
| 性质 | |
| 化学式 | C28H44O |
| 396.648 g·mol−1 g·mol⁻¹ | |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
参考文献
- Tomomi Morikawa, Masaharu Mizutani, Nozomu Aoki, Bunta Watanabe, Hirohisa Saga, Shigeki Saito, Akira Oikawa, Hideyuki Suzuki, Nozomu Sakurai, Daisuke Shibata, Akira Wadano, Kanzo Sakata, Daisaku Ohta. . The Plant Cell. 2006-04, 18 (4): 1008–1022 [2019-10-27]. ISSN 1040-4651. PMC 1425849
 . PMID 16531502. doi:10.1105/tpc.105.036012 (英语).
. PMID 16531502. doi:10.1105/tpc.105.036012 (英语). - 位于KEGG途径数据库的反应R07491
- 位于KEGG途径数据库的反应R07492
- Goad LJ, Holz GG, Beach DH. . Mol Biochem Parasitol. June 1985, 15 (3): 257–79. PMID 4033689. doi:10.1016/0166-6851(85)90089-1.
- Rodrigues JC, Attias M, Rodriguez C, Urbina JA, Souza W. . Antimicrob Agents Chemother. February 2002, 46 (2): 487–99. PMC 127026
 . PMID 11796362. doi:10.1128/AAC.46.2.487-499.2002.
. PMID 11796362. doi:10.1128/AAC.46.2.487-499.2002.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.