 | |
| Clinical data | |
|---|---|
| Trade names | Fenocyclin, Surestrine, Surestryl |
| Other names | Diosynestrol; Fenocycline; Fenocyclin; Phenocyclin; RS-2874; Dehydrofolliculinic acid; cis-Bisdehydrodoisynolic acid 7-methyl ether; BDDA ME; NSC-56846; NSC-122041 |
| Routes of administration | By mouth |
| Drug class | Nonsteroidal estrogen |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
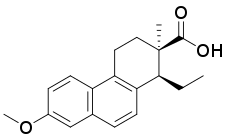
| Formula | C19H22O3 |
| Molar mass | 298.382 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Doisynoestrol (brand names Fenocyclin, Surestrine, Surestryl; former developmental code name RS-2874), also known as fenocycline, as well as cis-bisdehydrodoisynolic acid 7-methyl ether (BDDA ME), is a synthetic nonsteroidal estrogen of the doisynolic acid group that is no longer marketed.[1][2] It is a methyl ether of bisdehydrodoisynolic acid.[1] Doisynoestrol was described in the literature in 1945.[1] It has about 0.02% of the relative binding affinity of estradiol for the estrogen receptor.[3]
See also
References
- 1 2 3 Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 465–. ISBN 978-1-4757-2085-3.
- ↑ Herbai G (October 1971). "Separation of growth inhibiting potency from oestrogenicity in different weak oestrogenic drugs of various chemical structures". Acta Endocrinologica. 68 (2): 249–63. doi:10.1530/acta.0.0680249. PMID 5171465.
- ↑ Blair RM, Fang H, Branham WS, Hass BS, Dial SL, Moland CL, Tong W, Shi L, Perkins R, Sheehan DM (March 2000). "The estrogen receptor relative binding affinities of 188 natural and xenochemicals: structural diversity of ligands". Toxicol Sci. 54 (1): 138–53. doi:10.1093/toxsci/54.1.138. PMID 10746941.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.