菜籽多酚
菜籽多酚(英語:)是菜籽油中的酚类化合物[1][2],可在烘烤油菜种子时使芥子酸发生脱羧反应制得[3][4]。
| 菜籽多酚 | |
|---|---|
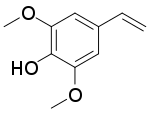 | |
| IUPAC名 4-Ethenyl-2,6-dimethoxyphenol | |
| 别名 | 2,6-Dimethoxy-4-vinylphenol 4-Vinyl-2,6-dimethoxyphenol 4-Vinylsyringol |
| 识别 | |
| CAS号 | 28343-22-8 |
| PubChem | 35960 |
| ChemSpider | 33074 |
| SMILES |
|
| 性质 | |
| 化学式 | C10H12O3 |
| 摩尔质量 | 180.2 g·mol−1 |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
参考文献
- Canolol: A Promising Chemical Agent against Oxidative Stress. Annia Galano, Misaela Francisco-Márquez and Juan R. Alvarez-Idaboy, J. Phys. Chem. B, 2011, 115 (26), pages 8590–8596, doi:10.1021/jp2022105
- 4-Vinyl-2,6-dimethoxyphenol (canolol) suppresses oxidative stress and gastric carcinogenesis in Helicobacter pylori-infected carcinogen-treated Mongolian gerbils. Cao X, Tsukamoto T, Seki T, Tanaka H, Morimura S, Cao L, Mizoshita T, Ban H, Toyoda T, Maeda H and Tatematsu M, Int J Cancer., 1 Apr 2008, 122(7), pages 1445-1454, PMID 18059022
- Antioxidant canolol production from a renewable feedstock via an engineered decarboxylase. Krista L. Morley, Stephan Grosse, Hannes Leischa and Peter C. K. Lau, Green Chem., 2013,n15, pages 3312-3317, doi:10.1039/C3GC40748A
- Isolation and Identification of a Potent Radical Scavenger (Canolol) from Roasted High Erucic Mustard Seed Oil from Nepal and Its Formation during Roasting. Kshitij Shrestha, Christian V Stevens, Bruno De Meulenaer, J. Agric. Food Chem., 2012, 60 (30), pp 7506–7512, doi:10.1021/jf301738y
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.