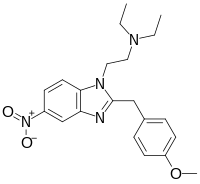
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C21H26N4O3 |
| Molar mass | 382.464 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Metonitazene is an analgesic compound related to etonitazene,[2][3] which was first reported in 1957,[4] and has been shown to have approximately 1000 times the potency of morphine by central routes of administration,[5] but if used orally it has been shown to have approximately 10 times the potency of morphine.[6]
Its effects are similar to other opioids such as fentanyl and heroin, including analgesia, euphoria, and sleepiness.[6][7] Adverse effects include vomiting, and respiratory depression that can potentially be fatal.[8] Because of high dependency potential and dangerous adverse effects it has never been introduced into pharmacotherapy. It is instead commonly used in the illicit manufacture of counterfeit-OxyContin opioid pills.
Legal status
In the United States, metonitazene is a Schedule I controlled substance under the Controlled Substances Act.
Metonitazene is not controlled under the 1971 Convention on Psychotropic Substances; however, in many countries possession or intent to sell for human consumption might be prosecuted under several analog acts.
See also
References
- ↑ Riksdagsförvaltningen. "Förordning (1992:1554) om kontroll av narkotika Svensk författningssamling 1992:1992:1554 t.o.m. SFS 2021:301 - Riksdagen". www.riksdagen.se (in Swedish).
- ↑ Vandeputte MM, Van Uytfanghe K, Layle NK, St Germaine DM, Iula DM, Stove CP (April 2021). "Synthesis, Chemical Characterization, and μ-Opioid Receptor Activity Assessment of the Emerging Group of "Nitazene" 2-Benzylbenzimidazole Synthetic Opioids". ACS Chemical Neuroscience. 12 (7): 1241–1251. doi:10.1021/acschemneuro.1c00064. hdl:1854/LU-8714061. PMID 33759494. S2CID 232337929.
- ↑ Ujváry I, Christie R, Evans-Brown M, Gallegos A, Jorge R, de Morais J, Sedefov R (April 2021). "DARK Classics in Chemical Neuroscience: Etonitazene and Related Benzimidazoles". ACS Chemical Neuroscience. 12 (7): 1072–1092. doi:10.1021/acschemneuro.1c00037. PMID 33760580. S2CID 232356192.
- ↑ Hunger A, Kebrle J, Rossi A, Hoffmann K (October 1957). "[Synthesis of analgesically active benzimidazole derivatives with basic substitutions]". Experientia. 13 (10): 400–401. doi:10.1007/bf02161116. PMID 13473817. S2CID 32179439.
- ↑ Hunger VA, Kebrle J, Rossi A, Hoffmann K (1960). "Benzimidazol-Derivate und verwandte Heterocyclen III. Synthese von 1-Aminoalkyl-2-nenzyl-nitro-benzimidazolen". Helvetica Chimica Acta. 43 (4): 1032–1046. doi:10.1002/hlca.19600430412.
- 1 2 Bromig G (October 1958). "[New powerful analgetics and their clinical testing]". Klinische Wochenschrift. 36 (20): 960–963. doi:10.1007/bf01486702. PMID 13612082. S2CID 1023209.
- ↑ Krotulski AJ, Papsun DM, Walton SE, Logan BK (October 2021). "Metonitazene in the United States-Forensic toxicology assessment of a potent new synthetic opioid using liquid chromatography mass spectrometry". Drug Testing and Analysis. 13 (10): 1697–1711. doi:10.1002/dta.3115. PMID 34137194. S2CID 235460764.
- ↑ Montanari E, Madeo G, Pichini S, Busardò FP, Carlier J (August 2022). "Acute Intoxications and Fatalities Associated With Benzimidazole Opioid (Nitazene Analog) Use: A Systematic Review". Therapeutic Drug Monitoring. 44 (4): 494–510. doi:10.1097/FTD.0000000000000970. PMID 35149665. S2CID 246776288.