 | |
| Clinical data | |
|---|---|
| Other names | 17α-Ethynylestradiol 3-O-sulfamate; J1028; EEMATE; EE2MATE; 17α-Ethynylestra-1,3,5(10)-triene-3,17β-diol 3-sulfamate |
| Routes of administration | By mouth[1] |
| Drug class | Estrogen; Estrogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| Chemical and physical data | |
| Formula | C20H25NO4S |
| Molar mass | 375.48 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
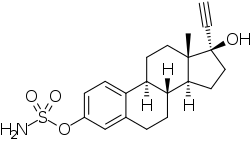
Ethinylestradiol sulfamate (developmental code name J1028), or 17α-ethynylestradiol 3-O-sulfamate, is a synthetic estrogen and estrogen ester which was never marketed.[1][2][3] It is the C3 sulfamate ester of ethinylestradiol.[1] The drug shows considerably improved oral estrogenic potency (uterotrophic) relative to ethinylestradiol in rats but without an increase in hepatic estrogenic potency.[1][3] Related compounds like ethinylestradiol N,N-diethylsulfamate (J271) and ethinylestradiol pyrrolidinosulfonate (J272) have also been developed, and have similar properties in animals.[4] However, the closely related compound estradiol sulfamate (E2MATE) failed to show estrogenic activity in humans, which is due to the fact that it is additionally a highly potent inhibitor of steroid sulfatase and prevents its own bioactivation into estradiol.[5]
See also
References
- 1 2 3 4 Elger W, Schwarz S, Hedden A, Reddersen G, Schneider B (December 1995). "Sulfamates of various estrogens are prodrugs with increased systemic and reduced hepatic estrogenicity at oral application". The Journal of Steroid Biochemistry and Molecular Biology. 55 (3–4): 395–403. doi:10.1016/0960-0760(95)00214-6. PMID 8541236. S2CID 31312.
- ↑ US 6080735, Schwarz S, Elger W, Siemann HJ, Reddersen G, Schneider B, "Estra-1,3,5(10)-trien derivatives, processes for their preparation and pharmaceutical compositions containing these compounds", issued 27 June 2000, assigned to Sterix Ltd
- 1 2 Stella VJ (12 March 2007). "Prodrug Approaches to Enhancing the Oral Delivery of Poorly Permeable Drugs". In Stella V, Borchardt R, Hageman M, Oliyai R, Maag H, Tilley J (eds.). Prodrugs: Challenges and Rewards. Springer Science & Business Media. p. 58. ISBN 978-0-387-49782-2.
- ↑ Elger W, Palme HJ, Schwarz S (April 1998). "Novel oestrogen sulfamates: a new approach to oral hormone therapy". Expert Opinion on Investigational Drugs. 7 (4): 575–589. doi:10.1517/13543784.7.4.575. PMID 15991994.
- ↑ Elger W, Wyrwa R, Ahmed G, Meece F, Nair HB, Santhamma B, et al. (January 2017). "Estradiol prodrugs (EP) for efficient oral estrogen treatment and abolished effects on estrogen modulated liver functions". The Journal of Steroid Biochemistry and Molecular Biology. 165 (Pt B): 305–311. doi:10.1016/j.jsbmb.2016.07.008. PMID 27449818. S2CID 26650319.